Discussion Overview
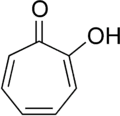
The discussion centers on the aromaticity of Tropolone, exploring the criteria for aromatic compounds and the specific structural characteristics of Tropolone that may or may not align with these criteria. Participants examine the implications of p-orbitals in the context of aromaticity, as well as potential experimental methods to test the aromatic nature of Tropolone.
Discussion Character
- Debate/contested
- Technical explanation
- Conceptual clarification
Main Points Raised
- One participant questions why Tropolone is considered aromatic, specifically regarding the presence of p-orbitals in the carbon atom connected to oxygen with a double bond.
- Another participant emphasizes the importance of Hückel's rule, noting that Tropolone meets the criteria of having 6 pi electrons in a conjugated, cyclic system.
- There is a discussion about the definition of whether a carbon atom "has" a p-orbital, with a suggestion to consider experimental methods for testing aromaticity.
- One participant proposes using NMR Spectroscopy as a potential method to evaluate the aromatic nature of Tropolone.
- A participant acknowledges a misunderstanding regarding the presence of p-orbitals due to the pi bond between carbon and oxygen, indicating a realization about the structure of pi bonds in Tropolone.
Areas of Agreement / Disagreement
Participants express differing views on the definition and implications of p-orbitals in relation to aromaticity, and there is no consensus on the understanding of Tropolone's aromaticity. The discussion remains unresolved regarding the experimental validation of its aromatic nature.
Contextual Notes
Limitations include potential ambiguities in definitions of p-orbitals and aromaticity, as well as the need for clarity on the structural characteristics of Tropolone that contribute to its classification.