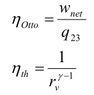Discussion Overview
The discussion revolves around the thermodynamic principles governing various engine cycles, specifically the Otto, Diesel, and Rankine cycles. Participants explore concepts such as the ideal gas law, the relationship between pressure, volume, and temperature, and the conditions under which certain equations apply, particularly in adiabatic processes.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
- Homework-related
Main Points Raised
- Some participants question whether the relationship PV/T=constant applies to all processes in all cycles, noting that it holds for ideal gases under certain conditions.
- There is confusion regarding the equation PV^γ=constant, with participants seeking clarification on its derivation and applicability, particularly in adiabatic processes.
- One participant asserts that explosions and shock waves do not follow the ideal gas law, indicating limitations of the law in non-equilibrium situations.
- Participants discuss the distinction between isothermal and adiabatic processes, with some asserting that PV is constant in isothermal processes while PV^γ is constant in adiabatic processes.
- There is a query about the definitions and differences between two types of efficiency (η(Otto) and η(thermal)), with participants expressing confusion over why two efficiencies exist.
- One participant raises a question about the calculation of power in an ideal Rankine cycle, suggesting that knowledge of P, V, T, and other parameters is necessary for determining power output.
Areas of Agreement / Disagreement
Participants express differing views on the applicability of the ideal gas law and the conditions under which various equations hold. There is no consensus on the definitions of efficiencies or the calculations related to the Rankine cycle, indicating ongoing debate and uncertainty.
Contextual Notes
Limitations include the dependence on ideal gas assumptions, the need for clarity on definitions of efficiency, and unresolved questions regarding the calculation of power in engine cycles.