Discussion Overview
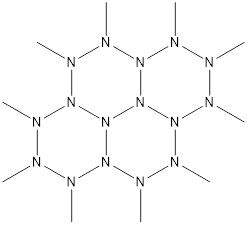
The discussion revolves around the theoretical possibility of a polymer structure involving N-methyl groups and nitrogen atoms. Participants explore the bonding characteristics, potential stability, and related compounds, focusing on theoretical and experimental aspects of nitrogen chemistry.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
Main Points Raised
- One participant questions the theoretical feasibility of a polymer structure with N-methyl groups, noting that while it does not violate elementary bonding rules, the strength difference between C-C and N-N bonds may lead to fragility.
- Another participant references the strong triple bond in elemental nitrogen, suggesting that this bond's characteristics dominate nitrogen chemistry and may influence the stability of proposed structures.
- A different participant discusses theoretical work on nitrogen allotropes, indicating that while hexagonal nitrogen could theoretically exist, it would likely be extremely explosive and challenging to synthesize.
- One participant mentions the creation of fluorescent nanodots of carbon nitride, suggesting a potential pathway for polymerization through specific conditions involving diazine or unsaturated amines.
Areas of Agreement / Disagreement
Participants express a range of views on the feasibility and stability of the proposed polymer structure, with no consensus reached on its theoretical possibility or practical implications. Multiple competing perspectives on nitrogen chemistry and polymerization methods are present.
Contextual Notes
Limitations include the dependence on specific bonding characteristics and the unresolved nature of the proposed polymer's stability and synthesis conditions.