Discussion Overview
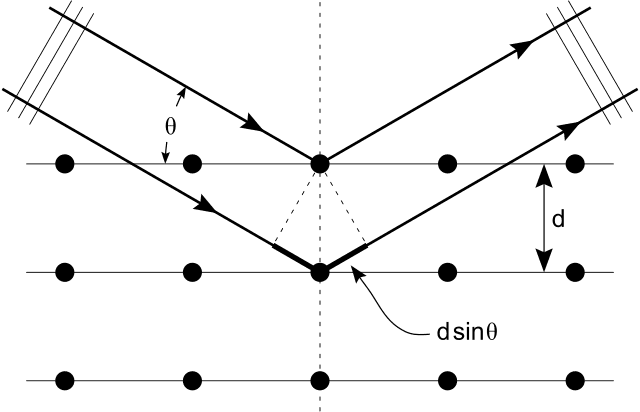
The discussion centers around the scattering of X-rays and the reasoning behind the path difference described by the formula 2dsinθ. Participants explore the principles of interference theory, particularly in the context of X-ray diffraction and scattering angles.
Discussion Character
- Exploratory
- Technical explanation
- Conceptual clarification
- Debate/contested
Main Points Raised
- One participant questions the reasoning behind the path difference of 2dsinθ and whether other scattering angles could exist.
- Another participant explains that the interference pattern created by a large number of sources in phase leads to specific angles for maxima, as determined by the wavelength and spacing, reinforcing the 2dsinθ relationship.
- It is noted that while atoms scatter at all angles, the intensity of the scattered X-rays has maxima at specific angles, and the diagram used is more of an aid for finding Bragg's formula rather than a complete depiction of the scattering process.
- A reference to Laue's theory of X-ray diffraction is made, suggesting it does not assume a specific scattering direction initially and applies to various scattering processes.
- Participants mention that similar calculations are applicable in other fields, such as radio engineering and ultrasound technology.
- One participant reiterates the importance of phase coherence among scatterers to achieve the desired scattering direction, emphasizing the need for a steady phase tilt across the row of scatterers.
Areas of Agreement / Disagreement
Participants express differing views on the interpretation of scattering angles and the completeness of the models discussed. There is no consensus on the necessity of specific scattering angles or the sufficiency of the explanations provided.
Contextual Notes
Some limitations in the discussion include the dependence on specific definitions of scattering angles and the assumptions made regarding phase coherence among scatterers. The discussion does not resolve these aspects.