- #1
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
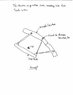
How to get reactions from given structure
In summary, the structure is statically indeterminate and assumptions must be made to calculate the reactions.
Engineering news on Phys.org
- #2
berkeman
Mentor
- 67,041
- 19,864
prelude1234 said:Can anyone tell me how to find the reactions of this structure? If not by summing forces and moments, then how? I'm thinking it's indeterminate and assumptions have to be made.
Welcome to the PF.
What is the context of the question? Is it for school?
- #3
prelude1234
- 9
- 0
No, this is a problem I'm having at work (aerospace) that I can't figure out.
- #4
nvn
Science Advisor
Homework Helper
- 2,128
- 32
prelude1234: Your structure is currently unstable, because it has no supports (also called constraints). A static structure must be constrained to its surroundings. In your diagram, please show the http://upload.wikimedia.org/wikipedia/en/c/cb/Simple_cantilevers_%28comparison%29.svg" (supports, boundary conditions) for your structure.
Last edited by a moderator:
- #5
- #6
nvn
Science Advisor
Homework Helper
- 2,128
- 32
prelude1234: It currently seems your structure is pinned to ground at every joint, although we are not 100 % sure, because you did not use the standard (clear) constraint http://upload.wikimedia.org/wikipedia/en/c/cb/Simple_cantilevers_%28comparison%29.svg" for connections pinned to ground. Also, one normally labels joints with capital letters, so they can be identified. And dimensions are usually necessary.
If all of your joints are pinned to ground, then you have four independent structures; and I think each structure is statically indeterminate, unless you assume one of the two pins on each member is a roller (?).
If all of your joints are pinned to ground, then you have four independent structures; and I think each structure is statically indeterminate, unless you assume one of the two pins on each member is a roller (?).
Last edited by a moderator:
- #7
- #8
Shockman
- 9
- 0
Hi
If Joint 1 and 3 not rigid, then the structure is not overconstrained so you do not need to remove pins.
If Joint 1 and 3 not rigid, then the structure is not overconstrained so you do not need to remove pins.
- #9
Studiot
- 5,440
- 9
Hello prelude, what company do you work for?
- #10
prelude1234
- 9
- 0
I am a contract Design Engineer with a major aerospace company. Why do you ask?
- #11
Studiot
- 5,440
- 9
Are other details in this company to the same standard?
I am bearing in mind that the application has potential public safety (airworthyness) involved.
You have stated that the structure is both rigid and pinned. This is incompatible.
nvn asked for a proper description of the problem, I do not see this in your second post.
Please re-present it using appropriate mechanics terms; it is impossible to determine anything from what you have presented so far.
I am bearing in mind that the application has potential public safety (airworthyness) involved.
You have stated that the structure is both rigid and pinned. This is incompatible.
nvn asked for a proper description of the problem, I do not see this in your second post.
Please re-present it using appropriate mechanics terms; it is impossible to determine anything from what you have presented so far.
- #12
AlephZero
Science Advisor
Homework Helper
- 6,982
- 298
Why do you assume it is a "detail"? For all you know, it might be the first sketch of a conceptual idea.Studiot said:Are other details in this company to the same standard?
Aerospace companies design lots of items that have nothing to do airworthiness, or public safety.I am bearing in mind that the application has potential public safety (airworthyness) involved.
There is nothing incompatible in having an (effectively) rigid structure pinned to something else (which may or may not be rigid). There may be good reasons for having an apparently redundant arrangement of pins - for example to deal with failure scenarios.You have stated that the structure is both rigid and pinned. This is incompatible.
The way I interpreted the OP was that this was a "rigid block" connected to something else with several pins, with a load applied at the CG of the block. The lines were just to define the geometric location of the pins. (But hey, what do I know, I just work in the aerospace industry...)
But given the later posts, I'm less sure about what it really means.
- #13
- #14
Studiot
- 5,440
- 9
Alephzero, the floor ( or roof? ) is all yours.
- #15
Shockman
- 9
- 0
Hi
This (your last drawing) is exactly the system like your drawing in the FEM system. No overconstrains.
First you calculate the forces in rod 1-2 and rod 1-3 (take joint 3 as pinned and remove rod 3-4 and 3-5.), so you get the forces in joint 3.
Secound you can calulate forces in rod 3-4 and 3-5 . (Remove rod 1-2 and rod 1-3, joint 3 is now not pinned)
If you are unsure, i can drop for you the detailed calculations.
This (your last drawing) is exactly the system like your drawing in the FEM system. No overconstrains.
First you calculate the forces in rod 1-2 and rod 1-3 (take joint 3 as pinned and remove rod 3-4 and 3-5.), so you get the forces in joint 3.
Secound you can calulate forces in rod 3-4 and 3-5 . (Remove rod 1-2 and rod 1-3, joint 3 is now not pinned)
If you are unsure, i can drop for you the detailed calculations.
Attachments
Last edited:
- #16
prelude1234
- 9
- 0
Thanks for the replies. I finally figured out the reactions by hand and simplifying the structure further, i.e. a simple dual triangle truss.
1. How can I predict the reaction products from a given chemical structure?
There are several methods for predicting reaction products from a given chemical structure. One approach is to use reaction databases or software programs that use known reaction rules and mechanisms to suggest potential products. Another method is to use retrosynthesis, which involves working backwards from the desired product to determine the necessary starting materials and reactions. Additionally, having a strong understanding of organic chemistry principles and reaction mechanisms can also aid in predicting products.
2. What factors influence the reactivity of a molecule?
The reactivity of a molecule can be influenced by a variety of factors, including functional groups, steric hindrance, electronic effects, and solvent effects. Functional groups with high electron density, such as alcohols and amines, tend to be more reactive. Steric hindrance, or the hindrance caused by bulky groups, can decrease reactivity by making it more difficult for reactants to come into contact with each other. Electronic effects, such as inductive and resonance effects, can also influence reactivity by altering the distribution of electrons in a molecule. Finally, the choice of solvent can also impact reactivity by affecting the stability of the reactants and products.
3. How can I determine the mechanism of a reaction?
Determining the mechanism of a reaction involves studying the individual steps or elementary reactions that make up the overall reaction. This can be done experimentally by monitoring the rate of reaction under different conditions or by using techniques such as kinetic isotope effects. Computational methods, such as density functional theory, can also be used to model and predict reaction mechanisms. Additionally, having a strong understanding of reaction mechanisms and their underlying principles can aid in determining the mechanism of a reaction.
4. What are some common types of reactions and their characteristics?
Some common types of reactions include substitution, elimination, addition, and oxidation-reduction reactions. Substitution reactions involve the replacement of a functional group or atom with another, while elimination reactions involve the removal of a functional group or atom to form a double bond. Addition reactions involve the addition of atoms or groups to a double or triple bond. Oxidation-reduction reactions, also known as redox reactions, involve the transfer of electrons between reactants. Each type of reaction has its own characteristic mechanism and can be further classified based on the functional groups or atoms involved.
5. How can I predict the stereochemistry of a reaction product?
Predicting the stereochemistry of a reaction product involves considering the chirality of the reactants and the mechanism of the reaction. If the reactants are chiral, the product will likely also be chiral. The mechanism of the reaction can then provide clues as to the stereochemistry of the product. For example, if the reaction follows an SN1 mechanism, the product will likely be a racemic mixture. If the reaction follows an SN2 mechanism, the product will likely have inverted stereochemistry. Additionally, computational methods can also be used to predict the stereochemistry of a product.
Similar threads
-
Mechanical Engineering
- Replies
- 2
- Views
- 953
-
Mechanical Engineering
- Replies
- 7
- Views
- 887
-
Mechanical Engineering
- Replies
- 12
- Views
- 4K
-
Mechanical Engineering
- Replies
- 4
- Views
- 1K
- Replies
- 1
- Views
- 1K
-
Mechanical Engineering
- Replies
- 6
- Views
- 3K
-
Mechanical Engineering
- Replies
- 5
- Views
- 30K
-
Mechanical Engineering
- Replies
- 11
- Views
- 2K
-
Mechanical Engineering
- Replies
- 7
- Views
- 1K
-
Mechanical Engineering
- Replies
- 1
- Views
- 1K
Share: