Mixing Definition and 282 Threads
-
Y
Mixing 18650 and 21700 cells in same parallel pack in battery pack ok?
I'm doing a 3S3P pack. My box can hold 9 18650s. It can also hold 3 18650s and 6 21700s. Is it ok to mix 18650s and 21700s in the same p pack? I would use the same 21700s and the same 18650s in each p pack. I would arrange them to maximize capacity. So say 18650 cells are Model A cells (making...- YoshiMoshi
- Thread
- Battery Cell Mixing
- Replies: 15
- Forum: Electrical Engineering
-
B
Undergrad Increasing tone while mixing sugar in water
I make Hummingbird feed by mixing 1/4 cup granulated sugar into 1 cup of water. I use a small glass pyrex measuring cup with one cup of water measured. It's heated for 1 minute in a microwave and 1/4 cup of sugar is added (no red dye!). I then vigorously stir the sugar and water using a metal...- billr
- Thread
- Mixing Sugar Water
- Replies: 10
- Forum: Other Physics Topics
-

Mixing two gases in an isolated system and calculating final p and T
Hey everyone, I have an attempt at fully solving this problem (my final pressure is ##p_f = 5373,64 hPa##, final temp. is ##T_f = 303,15K = 30C##), but this exercise confuses me very much. First, I have not used the masses in my calculations and I'm pretty sure my prof. accidentally copypasted...- chocopanda
- Thread
- Final Gas Gases Isolated system Mixing System Thermodyamics
- Replies: 4
- Forum: Introductory Physics Homework Help
-

Mixing two ideal gases with different V, T at constant pressure
To be honest, thermodynamics is really not my strong suit and I get confused when and how to apply formulas. My thought process is as follows: - there are two ideal gases (ideal gas law applies) - the pressure remains constant (isobaric process), so p1= p2 = p - I imagine there being two...- chocopanda
- Thread
- Constant Constant pressure Gases Ideal gas law Ideal gases Mixing Pressure Thermodynamics
- Replies: 16
- Forum: Introductory Physics Homework Help
-

Undergrad How Do You Calculate Equilibrium Conditions After Mixing Two Different Gases?
Consider that I have 2 adiabatic containers, one with a monoatomic gas at pressure P1, volume V1 and temperature T1 and another with pressure P2, volume V2, temperature T2. If I open a valvule and mix the two gases, how do I calculate the equilibrium pressure and temperature? I know the final...- jaumzaum
- Thread
- Gases Mixing
- Replies: 3
- Forum: Thermodynamics
-

Undergrad Evaluating the quark neutrino mixing matrix
The mixing of the 3 generations of fermions are tabulated into the CKM matrix for quarks: $$ \begin{bmatrix} c_{12}c_{13} & s_{12}c_{13} & s_{13}e^{-i\sigma_{13}} \\ -s_{12}c_{23}-c_{12}s_{23}s_{13}e^{i\sigma_{12}} & c_{12}c_{23}-s_{12}s_{23}s_{13}e^{i\sigma_{13}} & s_{23}c_{13} \\...- James1238765
- Thread
- Matrix Mixing Neutrino Quark
- Replies: 3
- Forum: Quantum Physics
-
S
High School Graphical representation of the weak mixing angle
The graphical pattern of particles in the weak hypercharge and weak isospin plane, visible in this wiki page, shows the mixing angle between the Yw and Q axes. Actually , from the weak hypercharge (-2) of a right-handed electron and its electric charge (-1), one obtains an angle Pi/3, not the...- Sandglass
- Thread
- Angle Mixing Representation Weak
- Replies: 12
- Forum: High Energy, Nuclear, Particle Physics
-
T
Undergrad Temperature change due to mixing liquids, heating and heat losses
Hello, I am stuck how to proceed with the equations below. The problem: A tank containing ##m_0## mass, with a temperature of ##T_0##, is to be filled with a total mass of ##m_1##, with the constant mass flow rate ##G## and temperature ##T_1## during ##t_p## seconds. ##T_0>T_1##. Other than the...- tjosan
- Thread
- Change Heat Heating Liquids Mixing Temperature Temperature change
- Replies: 8
- Forum: Thermodynamics
-
J
Undergrad How Can Laser Light Mixing Reveal Quantum Entanglement?
https://quantum.phys.lsu.edu/old-website/seminars/abstracts/Kaushik10.pdf I have discovered an experiment in the link above where you get NOON entangled states by mixing classical laser light with quantum light from SPDC. But I cannot understand the mathematics behind it. Can you explain it to...- James2018
- Thread
- Entanglement Experiments Laser Light Mixing
- Replies: 16
- Forum: Quantum Physics
-

Mixing between Neolithic Farmers and Mesolithic Hunter-Gatherers
I watching this YouTube video about Göbekli Tepe which suggests that hunter-gatherers may have been more capable of creating civilizations than we presumed and it got me wondering what hunter-gatherer / farmer relations were like. Apparently they were good...- Pythagorean
- Thread
- Mixing
- Replies: 5
- Forum: Biology and Medical
-

Can I make a rocket by mixing aluminum + iodine (+ water catalyst)?
Basically it's a well known reaction, many kids on youtube make it for the views: I am thinking, since AlI3 sublimes can it be put in say tungsten bottle (is this even a thing??) and then used as a rocket the same way Na+H2O or HCL+Al rockets are used - the gas escapes? This phase... -
R
Looking for basic theory on mixing of gases
Hi, In my situation, I am dealing with two ideal gases traveling down a pipe at the same bulk velocities. I am only considering a 1-D treatment at the moment and I would like to find out at which point the two gases should be considered a well mixed, homogeneous mixture. Would anyone know...- random_soldier
- Thread
- Gases Mixing Theory
- Replies: 4
- Forum: Mechanical Engineering
-

Is it possible to have an "Eternal reaction" by mixing the products?
So this is a very simple concept but I am not explaining it well: Let's say you have 4 reactants in 2 different reactions (apologies: the symbols I am using do NOT exist, other than the "C" symbol, I just can't find real elements so that I am using just alphabet symbols as an allegory to show... -
J
My first time mixing Kroll's reagent
I am going to etch some titanium and need to mix up a 100ml batch of Kroll's reagent ( 92ml distilled water, 6ml nitric acid and 2ml hydrofluoric acid. Do I add the nitric acid to the water first and then add the hydrofluoric or vice-versa? -

Departure enthelpy for mixture EOS = enthelpy of mixing?
Hi. I am well aware that if say, there are two components A and B, the ideal mixing enthalpy between those two components are: (1) hmix = zA hA + zB hB. If I want to get the real behavior of a pure component, I am also aware that; say for example, liquid enthalpy: (2) hL = Href + ∫CpigdT (from...- maistral
- Thread
- Mixing Mixture
- Replies: 2
- Forum: Materials and Chemical Engineering
-
A
Solving a mixing problem with a differential equation
Hello! First I tried modelling it like most mixing problems. $$ \frac{dA}{dt} = rate coming in - rate coming out $$ where dA is the volume and dt is the time rate coming in/out can be describe as; contrencation * flow rate. Now if we plug that all on $$ \frac{dA}{dt} = 35 * 0 -...- arhzz
- Thread
- Differential Differential equation Mixing
- Replies: 2
- Forum: Calculus and Beyond Homework Help
-

Mixing Newton's law with Adiabatic Process
I have attached an image showing the perimeters of the problem. I have included what I think is the solution, could someone please take a look and tell me if I am on the correct path, in the solution I am taking Joules as a common term to attempt to solve the question. The gas I have used is N...- TonyCross
- Thread
- Adiabatic Adiabatic process Law Mixing Newton's law Process
- Replies: 11
- Forum: Introductory Physics Homework Help
-

How do I get the Convection Coefficient for steam in a jacketed vessel?
Please could someone point me in the direction of an equation/ book I can use to obtain the convection coefficient for steam in a heating jacket. Steam is at 1 bar pressure. I think I need to use a dimensionless number. Thanks- Will26040
- Thread
- Chemical engineering Coefficient Conduction Convection Heat transfer Mixing Steam Vessel
- Replies: 3
- Forum: Materials and Chemical Engineering
-
Z
Undergrad Why does mixing blue and yellow create green?
I am curious about how mixing paints works. If I mix an ideal paint A and an ideal paint B what is the product mixture C? Is it (Option 1) a mixture absorbs all the range A absorbs and absorbs all the range B absorbs OR is it (Option 2) a mixture with original A and B pigments therefore some... -
M
Undergrad What is the purpose of the Prandtl Mixing Length for turbulent flow?
Hi, I was recently learning about turbulent boundary layers and came across the 'Prandtl mixing length'. I am struggling to understand what the concept is and what its purpose is. I would appreciate any help or guidance of where I can look to gain a better understanding. The information I...- Master1022
- Thread
- Flow Length Mixing Turbulent flow
- Replies: 2
- Forum: Thermodynamics
-

Helmholtz free energy for mixing?
Hi, I have been reading a few literature regarding excess Helmholtz energy and I encountered this definition from the paper of Wong and Sandler (apparently, from the mixing rule used in a EOS): In particular, the ones in the red boxes. How did these equations come into being? I tried to look...- maistral
- Thread
- Energy Free energy Helmholtz Helmholtz free energy Mixing
- Replies: 1
- Forum: Materials and Chemical Engineering
-

Undergrad Experimental test of the Mixing Paradox
Can anybody point to an experiment that shows that the total entropy increases if two different gas mix at constant pressure and temperature, whereas if two volumes of identical gases mix the total entropy doesn't change?- Philip Koeck
- Thread
- Experimental Mixing Paradox Test
- Replies: 13
- Forum: Thermodynamics
-
R
Graduate Gibbs "Paradox" and the Entropy of mixing
(not a paradox nowadays, but it was an issue for years) https://en.m.wikipedia.org/wiki/Gibbs_paradox It's not a question about a formula. I don't understand the motivation in physics to claim Gibbs mixing "paradox", the discontinuity point. What bothers the physicist to ask for a continuous...- rentier
- Thread
- Entropy Gibbs Mixing Paradox
- Replies: 3
- Forum: Thermodynamics
-

Chemistry What precipitates form by mixing 2 saturated solutions?
At first I thought it was C. A few sources agree with me. The book says D is correct. Here was my reasoning for C: With both salts having a formula of MX3, their [OH-]'s can be compared according to their Ksp values. Tl(OH)3 has a smaller Ksp, so it has a smaller [OH-] at saturation than...- sneakycooky
- Thread
- Form Ksp Mixing Saturation
- Replies: 2
- Forum: Biology and Chemistry Homework Help
-

Controlling two valves with different flow-rates to achieve mixing
Guys, I hope you guys are keeping well. I have been working on developing a Compact Dispensing Station for my CNC Machine & Parts Washer where coolant is mixed with water in different concentrations & volumes as per the requirement which will vary on a day-to-day basis (not the concentration...- kunalvanjare
- Thread
- Mixing Valves
- Replies: 23
- Forum: Materials and Chemical Engineering
-
E
Mixing of two polymer particles heated above Tg
I am wondering if two polymer particles, of same kind, heated above Tg can be mixed, over time, if they are squeezed together. By mixing I mean that the surfaces of the two particles will mix into one after they are squeezed together. For simplicity let's assume that the polymer particles are...- Excom
- Thread
- Mixing Particles Polymer
- Replies: 6
- Forum: Materials and Chemical Engineering
-

MHB Regarding mixing time of a Markov chain
Let $X=X_0, X_1, X_2, \ldots$ be a Markov chain on a finite state space $S$, and let $P$ denote the transition matrix. Assume that there is an $\varepsilon>0$ such that whenever $\mu_0$ and $\nu_o$ are point distributions on $S$ (in other words, $\mu_0$ and $\nu_0$ are Direac masses) we have...- caffeinemachine
- Thread
- Chain Markov chain Mixing Time
- Replies: 2
- Forum: Set Theory, Logic, Probability, Statistics
-

[Differential equations] Mixing problem.
v(t)=600+(9-6)t =600+3t 1500=600+3t therefore t=300 hrs when tank is full Cin=1/5(1 + cost) ds/dt=Rate in - rate out = CinRin - count*S(t)/V(t) =1/5(1 + cost)*9 - 6*S(t)/(600+3t) S(0)=5 ib Solving the first order linear ODE we get: https://www.desmos.com/calculator/l7iixzgyll therefore...- Muskyboi
- Thread
- Differential equations Mixing
- Replies: 2
- Forum: Calculus and Beyond Homework Help
-
K
Gibbs' theorem and partial molar volume
In the chemical engineering text of Smith, VanNess, and Abbott, there is a section on partial molar volume. It states that Gibbs theorem applies to any partial molar property with the exception of volume. Why is volume different? In other words, when evaluating the partial molar volume of a...- kayan
- Thread
- Chemical engineering Gibbs Mixing Partial Theorem Thermodynamics Volume
- Replies: 4
- Forum: Materials and Chemical Engineering
-

How can I achieve homogeneous in-line mixing of a Coolant with water?
Hello guys, This is for a new product I am designing, which involves topping-up or refilling the liquid into a CNC Machine or Part Washer. I have to mix about a Litre of the chemical (alkaline.. pH in the range of 8-12) with about 50 Litres of water. The water will be fed under gravity through...- kunalvanjare
- Thread
- Coolant Homogeneous Mixing Water
- Replies: 1
- Forum: Materials and Chemical Engineering
-
High School Mixing two discrete audio signals
Suppose I have two audiofiles in 16 bit PCM, both recorded on a level that, except for the noise and distortion, is maximally recorded, or that the maximum recording level results in the maximum PCM level. So, the signal is recorded on the maximum level such that there is no clipping. If we...- entropy1
- Thread
- Audio Discrete Mixing Pcm Signal Signals
- Replies: 13
- Forum: General Math
-
L
Mixing Substances: Engineering Solutions for Dilution Challenges by Lee
Hello, my name is Lee. I am a design and electrical engineer and I am involved in a project where I have developed a piece of equipment that mixes substances, primarily two liquids. My equipment is a rotational shaking machine. The liquids are put into a vessel which is then shook over a number... -
S
Mixing Fluids in Flow: How to Achieve a Fixed Concentration | Sanatan
Hello, I am trying to mix two fluids which are in flow. To be exact, I am trying to mix a concentrated mineral solution to an outlet of my RO filter. Is there a method to mix the two fluids to get a fixed concentration of the mixed solution ? Regards, Sanatan- sanatan
- Thread
- Flow Fluids Mixing
- Replies: 4
- Forum: Materials and Chemical Engineering
-

High School Can He be used as a safer alternative to H in blimps?
Saw the saddest thing in the world today: party balloons peeking out over the top of a dumpster. I started thinking about the buoyancy of H versus its flammability. H is 6-7 times more buoyant than He. It would be more more economical to use H if we could eliminate the explody bit. If you can...- DaveC426913
- Thread
- Mixing
- Replies: 24
- Forum: Classical Physics
-
T
Change in entropy when mixing cold and hot water
I tried following: $$ dS_{\text{total}} = |\frac{dQ}{T_c}| |\frac{dQ}{T_H}| $$ where ## T_h ## is temperature of hot water and ## T_c ## is temperature of cold water. Coefficient for water wasn't provided in the assignment so i used following value c = 4190 J/kgK. $$ dS_{\text{total}} =...- tuki
- Thread
- Change Cold Entropy Hot Mixing Water
- Replies: 12
- Forum: Introductory Physics Homework Help
-

Quantum Gases - Mixing of 3He and 4He
Homework Statement Consider a solution in which 99% of the atoms are 4He and 1% are 3He. Assuming that the 3He atoms behave as an ideal gas of spin-1/2 particles determine the Fermi energy of the 3He atoms. You may assume that one mole of 4He occupies a volume of 28 cm3.Homework Equations EF =...- i_hate_math
- Thread
- Fermi-dirac statistics Gases Mixing Quantum Statistical physics
- Replies: 1
- Forum: Advanced Physics Homework Help
-
C
How does the phase noise of the LO effect IF accuracy?
Hi, I have a roughly 1.1 GHz signal to be downconverted to 100 MHz by mixing it with a 1 GHz local oscillator. I am not sure how to choose the performance of the LO. In particular: let's assume the LO has a jitter of 100 fs rms. At 1 GHz this corresponds to a frequency error of 100 kHz. Does...- csopi
- Thread
- Accuracy Mixing Noise Phase
- Replies: 4
- Forum: Electrical Engineering
-
K
High School Additive colour theory and "traditional" colour mixing
Hi folks. I am trying to reconcile the additive colour theory of light with more traditional grade school colour mixing. For example, we all know that blue paint plus yellow paint makes green paint. How do we explain this from the perspective of light? So the yellow paint appears yellow because...- keroberous
- Thread
- Colour Mixing Theory
- Replies: 8
- Forum: Optics
-

Do we assume the mixing chamber has an outlet?
Homework Statement Hot and cold streams of a fluid are mixed in a rigid mixing chamber. The hot fluid flows into the chamber at a mass flow rate of 5 kg/s with an energy in the amount of 150 kJ/kg. The cold fluid flows into the chamber with a mass flow rate of 15 kg/s and carries energy in the...- EastWindBreaks
- Thread
- Chamber Mixing Open system Thermodaynamics
- Replies: 3
- Forum: Introductory Physics Homework Help
-
G
Chemistry How to Convert a Number Concentration into Volume Mixing Ratio?
Homework Statement Convert a number concentration of 15 X 10^12 molecule/cm3 into volume mixing ratio at: a) 1 atm pressure, and 20 C temperature. b) 500 mbar pressure, and 253 K temperature. Using the Ideal Gas Law, calculate: a) Loshmidt’s number (# of molecules/cm3 of air, at standard...- Geo_Zegarra2018
- Thread
- Concentration Convert Mixing Ratio Volume
- Replies: 3
- Forum: Biology and Chemistry Homework Help
-

Measuring Torque on the Impeller Shaft of a Stirred Tank
Hi Everyone, I am working on a project which requires me to determine the torque on the impeller shaft of stirred tank. Currently as I see it, there are 2 viable options: To measure the torque in the shaft using a torque transducer such as this one. This is by far the most accurate means of...- runningman19
- Thread
- Fluid dynamics Measuring Mixing Shaft Tank Torque
- Replies: 10
- Forum: Materials and Chemical Engineering
-
C
Practical Fluid Mechanics: inversions & mixing
I'm trying to think through a situation, and develop some confidence I've got it right. The situation is a apartment building, about 60'/20m high, with a courtyard in the middle about 60'/20m square. In the middle of the courtyard, is an low exhaust stack, about 8' high and around 8' square...- CodeScience
- Thread
- Fluid Fluid mechanics Mechanics Mixing Practical
- Replies: 7
- Forum: General Engineering
-
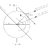
Undergrad How Can Fleet Mixing Optimize Crew Encounters in Regattas?
Dear all, we are organizing a regatta, there are 12 competing crews and we have six boats. Therefore the regatta is organized in so-called fleets, each consisting of two races at which six crews (using the six available boats) are participating. We assume to be able to race 10 to 15 fleets...- stockzahn
- Thread
- Mixing Statistics probability
- Replies: 15
- Forum: Set Theory, Logic, Probability, Statistics
-

Graduate 1D Convection Diffusion Equation - Inlet Mixing Effect
I have a working Matlab code solving the 1D convection-diffusion equation to model sensible stratified storage tank by use of Crank-Nicolson scheme (without εeff in the below equation). As indicated by Zurigat et al; there is an additional mixing effect having a hyperbolic decaying form...- HumanistEngineer
- Thread
- 1d Convection Diffusion Diffusion equation Inlet Mixing Storage
- Replies: 7
- Forum: Differential Equations
-
L
Solving Entropy of Mixing Problem in Physics
Hi again Physics Forums! Last time I was here, I was an undergrad student. Now I almost finished a PhD in quantum information and became a high school teacher. I've never properly learned thermodynamics. I'm now trying to get it on the same level, that I understand the other topics in classical...- Loro
- Thread
- Entropy Mixing
- Replies: 17
- Forum: Introductory Physics Homework Help
-
M
MHB Mixing with Common Drain: Mass of Salt in Two Tanks #46 Nagle
Mixing with a Common Drain. Two tanks, each holding 1 L of liquid, are connected by a pipe through which liquid flows from tank A into tank B at a rate of 3-a L/min (0<a<3). The liquid inside each tank is kept well stirred. Pure water flows into tank A at a rate of 3 L/min. Solution flows out of...- mathcoral
- Thread
- Differential Differential equations Fundamentals Mixing
- Replies: 6
- Forum: Differential Equations
-
Undergrad Quark mixing and energy conservation
We've recently been looking at the hadronic decays of the W boson. In this one example, we looked at possible decays for the W boson being produced near its resonance peak, meaning the centre of mass energy is sufficient to produce u,d,c,s & b quarks. However, because we're below the mass of...- Jezza
- Thread
- Conservation Energy Energy conservation Mixing Quark
- Replies: 4
- Forum: High Energy, Nuclear, Particle Physics
-
V
Mixing CP asymmetry calculation
Homework Statement Consider decay ##\bar{B^0_s}\rightarrow D^+_s \pi^-## and calculate its mixing-induces CP asymmetry. Homework Equations $$\xi_f^{s}=e^{i\theta_{M_{12}}} \frac{A(\bar{B^0_q}\rightarrow f)}{A(B^0_q \rightarrow f)}=\pm e^{-i\phi_s}\frac{...- Valeriia Lukashenko
- Thread
- Asymmetry Calculation Cp violation Mixing
- Replies: 3
- Forum: Advanced Physics Homework Help
-

Rotating magnetic field effect on aqueous mixing (opinions?)
This is question that can't really get a precise answer without significant modeling or experimentation, but I'm wondering what folks' intuition might be on an answer to the following setup + question: Suppose there is a steel tank made of steel pipe with walls about 0.25 cm thick, internal...- Irl495
- Thread
- Aqueous Electromagetic field Field Magnetic Magnetic field Magnetism Mixing Opinions Reactor design Rotating
- Replies: 4
- Forum: Materials and Chemical Engineering
-
M
Rushton Turbine Torque Calculation - D, H, Viscosity, RPM
Hi; In one of my projects I need a Rushton impeller to be used for mixing applications. However, these kinds of impellers are quite expensive(400$ for a 2inch impeller) if bought from laboratories equipment manufacturers. The price tag would be fine with me if only one blade was needed, but this...- Mechstudent
- Thread
- Mixing Torque Turbine
- Replies: 1
- Forum: Mechanical Engineering