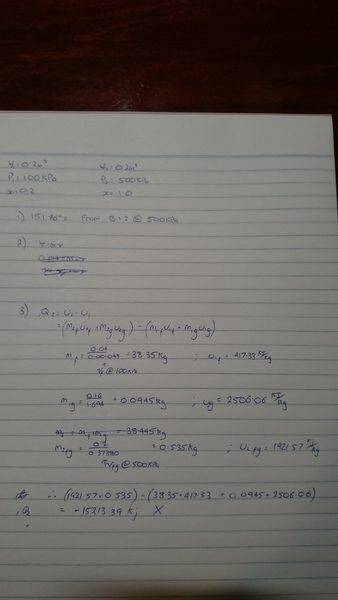Discussion Overview
The discussion revolves around a thermodynamics problem related to the First Law of Thermodynamics, specifically focusing on the calculations involving the mass and specific volumes of water and vapor in a tank at different states. Participants explore the application of steam tables and the implications of saturated and superheated states.
Discussion Character
- Homework-related
- Technical explanation
- Debate/contested
Main Points Raised
- One participant expresses confusion about the calculations for internal energy and the correct approach to finding the mass of saturated vapor at different states.
- Another participant suggests starting by determining the initial mass of water using steam tables.
- There is a discussion about the correct interpretation of steam quality and its relation to mass fractions versus volume fractions.
- Participants calculate specific volumes and total masses, but some calculations are challenged or corrected by others, indicating potential misunderstandings.
- One participant identifies a mistake in their understanding of the quality of the mixture, which leads to a resolution of their calculations.
- Concerns are raised about the specific volume of the mixture at certain pressures and temperatures, with references to the conditions for saturated versus superheated vapor.
- Final calculations are presented, showing the mass of liquid and vapor in the tank, along with a query about the application of the First Law in this context.
Areas of Agreement / Disagreement
Participants do not reach a consensus on all aspects of the problem, as there are multiple corrections and differing interpretations of the calculations and definitions involved. Some participants agree on certain calculations while others express confusion or propose alternative approaches.
Contextual Notes
Limitations include potential misunderstandings of steam table data, assumptions about the states of water and vapor, and the specific conditions under which calculations are made. The discussion reflects a progression of thought and refinement of ideas rather than a straightforward resolution.
Who May Find This Useful
Students and practitioners in thermodynamics, particularly those dealing with steam tables and the First Law of Thermodynamics, may find this discussion relevant.