Discussion Overview
The discussion revolves around the operation of batteries connected in series, specifically addressing the conditions under which chemical reactions occur within the batteries and the implications for current flow in a circuit. Participants explore the electrochemical processes involved and troubleshoot a scenario where no activity is observed in the circuit.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
Main Points Raised
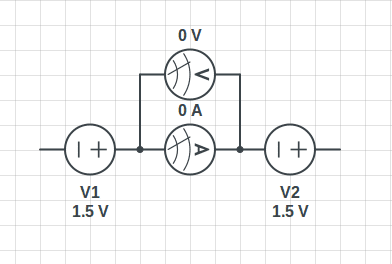
- Some participants express confusion about the path of electrons in batteries connected in series and why no current flows despite the electrodes being immersed in electrolytes.
- One participant suggests that a complete circuit path is necessary for current to flow, indicating that chemical reactions do not initiate until the circuit is closed.
- Another participant argues that electrochemical reactions begin as soon as the battery is assembled but typically reach an equilibrium state with no current until the circuit is completed.
- A detailed explanation of the electrochemical reactions occurring at the anode and cathode of a lead-acid battery is provided, describing the processes of oxidation and reduction that generate electricity when a load is applied.
- One participant questions the concept of initial equilibrium mentioned by another, seeking clarification and additional resources on the topic.
Areas of Agreement / Disagreement
Participants do not reach a consensus on the nature of the initial conditions for chemical reactions in batteries. There are competing views regarding the timing of when reactions begin and the necessity of a complete circuit for current flow.
Contextual Notes
Some statements rely on assumptions about the behavior of batteries and the definitions of equilibrium in electrochemical reactions, which are not fully resolved in the discussion.