Homework Help Overview
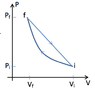
The discussion revolves around calculating the efficiency of a heat engine that operates through an adiabatic compression followed by a linear expansion. Participants express confusion regarding the efficiency calculation, particularly the relationship between work done and heat absorbed during the processes involved.
Discussion Character
- Exploratory, Assumption checking, Conceptual clarification
Approaches and Questions Raised
- Participants question whether the work (W) refers to the net work or the work done by the gas. There is discussion about how Q_H relates to the adiabatic process, with some suggesting that Q_H equals the negative of the net work. Others explore the implications of Q being zero during adiabatic steps and how that affects the efficiency calculation.
Discussion Status
Several participants have provided insights into the relationship between work and heat flow, particularly focusing on the first law of thermodynamics. There is an ongoing exploration of how to correctly identify Q_H and the work done during each part of the cycle. Multiple interpretations of the processes are being discussed, with no explicit consensus reached yet.
Contextual Notes
Participants note the complexity of determining heat flow during different phases of the engine cycle, particularly in distinguishing between heat absorbed and heat released. There is also mention of specific temperatures and pressures at various stages, which may influence the calculations but are not fully resolved in the discussion.