Homework Help Overview
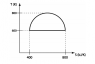
The discussion revolves around a thermodynamic cycle, specifically focusing on determining the direction of the cycle (clockwise or anticlockwise) and calculating the work done within the cycle. The context involves applying the first law of thermodynamics and understanding the relationship between temperature, entropy, and work in thermodynamic processes.
Discussion Character
- Exploratory, Conceptual clarification, Assumption checking
Approaches and Questions Raised
- Participants discuss the implications of the first law of thermodynamics and the significance of integral signs in the equations. There are attempts to clarify the relationship between the area under the T-dS plot and the work done, with some questioning the nature of the processes involved in the cycle.
Discussion Status
The conversation is ongoing, with participants exploring different interpretations of the cycle's direction and the calculation of work. Some guidance has been provided regarding the relationship between the area on the T-S diagram and work, yet there remains uncertainty about the exact calculations and assumptions regarding the processes represented.
Contextual Notes
Participants note the lack of specific information about the types of processes in the cycle, which complicates the calculation of work. There is also a discussion about the implications of the cycle's direction on the sign of the work done, highlighting the need for clarity on assumptions made in the analysis.