Discussion Overview
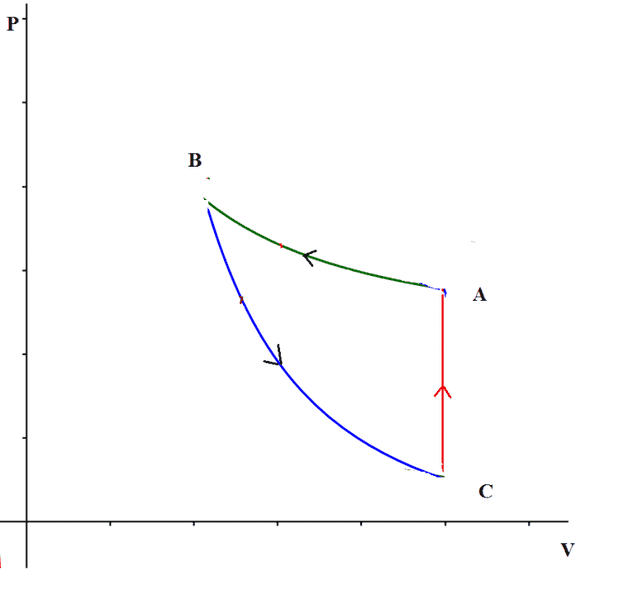
The discussion revolves around the work done by an ideal gas during a thermodynamic cycle represented on a P-V plane, specifically questioning whether the orientation of the cycle (clockwise or counterclockwise) determines the sign of the work done. Participants explore various scenarios involving different types of processes within the cycle, including isochoric and adiabatic processes.
Discussion Character
- Debate/contested
- Technical explanation
- Conceptual clarification
Main Points Raised
- Some participants assert that a clockwise cycle indicates positive work done by the gas, while a counterclockwise cycle indicates negative work, but this may not hold true in all cases.
- One participant presents a specific cycle involving two adiabatic processes and one isochoric process, arguing that the work can be positive even if the cycle is traveled counterclockwise.
- Another participant challenges this view, emphasizing that the work done by the gas during expansion and the work done on the gas during compression must be considered, suggesting that the area under the P-V curve dictates the sign of the work.
- There is a discussion about the heat flow during isothermal and isochoric processes, with some participants arguing that heat flow is not limited to the isochoric process and that it must be accounted for in the overall cycle.
- Concerns are raised regarding the implications of irreversible processes on the validity of the work calculations, with suggestions that the pressure at the piston must be considered for accurate work determination.
- One participant questions whether a cycle can exist with certain characteristics, leading to a broader inquiry about the conditions under which work and heat flow can be positive or negative.
Areas of Agreement / Disagreement
Participants express differing views on the relationship between cycle orientation and work sign, with no consensus reached. Some argue for the validity of the clockwise/counterclockwise rule, while others present counterexamples and challenge the generality of this rule.
Contextual Notes
Participants note that the discussion involves complexities related to irreversible processes and the assumptions made about heat flow during different types of thermodynamic processes. The implications of these factors on the overall analysis of work and heat in cycles remain unresolved.