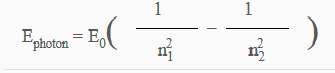Discussion Overview
The discussion revolves around the equations used to model atomic spectra, particularly questioning whether a general equation exists for all atoms or if it is limited to hydrogen and its isotopes. Participants explore the implications of quantum mechanics on atomic spectra and the complexity involved in deriving a universal formula.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
Main Points Raised
- Some participants assert that the equation discussed is specific to hydrogen and approximately applicable to Rydberg states.
- There is a question about whether atomic spectra are the same for all isotopes, leading to a discussion on isotope shifts.
- One participant mentions that the isotope shift occurs due to the motion of the nucleus affecting the wavefunction and energy levels of electrons.
- A formula for the isotope shift is presented, involving the reduced mass and energy levels.
- Another participant suggests that quantum mechanics introduces probabilities, complicating the derivation of a general result for multielectron atoms.
- In contrast, another participant argues that the lack of a general result is due to the complexity of the systems rather than the probabilistic nature of quantum mechanics.
Areas of Agreement / Disagreement
Participants generally agree that the equation is specific to hydrogen and that no general expression for all atoms has been derived. However, there is disagreement regarding the reasons for the absence of a universal formula, with differing views on the role of quantum mechanics and complexity.
Contextual Notes
Limitations include the lack of a comprehensive understanding of multielectron atoms and the dependence on specific conditions for isotopes. The discussion does not resolve the complexities involved in modeling atomic spectra for all atoms.