texaska
- 4
- 0
Hello,
i would like to simulate an ideal gas flow in a cube...the cube consists of small cubes lined next to each other..after warming up one small cube i should describe how is the heat spreading to the rest of the smaller cubes...using eulers equations for inviscid fluid motion 3D..
using the equation of state I count the mass of molecules inside one small cube,a=1m
V=1m3
P=100KPa
T=293.15K
r=8.314
n=PV/rT
n=41.0299mol
------------------------
i warm up one small cube to 293.25K,so the pressure will rise
now according to the equation for axes x
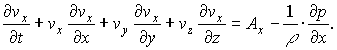
it should be something like
Vx_new-Vx+(Vx-V(x-1))*Vx=-(Px-P(x-1))/rho
where
Vx_new-Vx=local acceleration
(Vx-V(x-1))*Vx=konvective acceleration
the rest can be neglected cause it is laminar flow
----------------------------
as far as i know the molecules in the cube with higher pressure should move to cubes with lower pressure
------------------------------
the problem is that using this formula the molecules spread also from cubes with lower pressure to cube with higher pressure which is wrong..and i don t know how to twist the formula to prevent that..
-------------------------
so please if u have some experiences with modeling gas flow using there equations or if u see a mistake in my rewritten formula please let me know..
--------------------------
i would like to simulate an ideal gas flow in a cube...the cube consists of small cubes lined next to each other..after warming up one small cube i should describe how is the heat spreading to the rest of the smaller cubes...using eulers equations for inviscid fluid motion 3D..
using the equation of state I count the mass of molecules inside one small cube,a=1m
V=1m3
P=100KPa
T=293.15K
r=8.314
n=PV/rT
n=41.0299mol
------------------------
i warm up one small cube to 293.25K,so the pressure will rise
now according to the equation for axes x
it should be something like
Vx_new-Vx+(Vx-V(x-1))*Vx=-(Px-P(x-1))/rho
where
Vx_new-Vx=local acceleration
(Vx-V(x-1))*Vx=konvective acceleration
the rest can be neglected cause it is laminar flow
----------------------------
as far as i know the molecules in the cube with higher pressure should move to cubes with lower pressure
------------------------------
the problem is that using this formula the molecules spread also from cubes with lower pressure to cube with higher pressure which is wrong..and i don t know how to twist the formula to prevent that..
-------------------------
so please if u have some experiences with modeling gas flow using there equations or if u see a mistake in my rewritten formula please let me know..
--------------------------