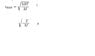Finding the Root Mean Square Speed of Methane at 50°C
Click For Summary
Homework Help Overview
The original poster seeks to find the root mean square speed of methane at a temperature of 50 degrees Celsius, expressing uncertainty about which formula to use for the calculation.
Discussion Character
- Exploratory, Conceptual clarification, Assumption checking
Approaches and Questions Raised
- Participants discuss two formulas related to the calculation, with questions about the variables involved, such as the meaning of R in one formula and the relevance of the second formula. There is also an inquiry about the molar mass of methane.
Discussion Status
Some participants have provided clarifications regarding the formulas and constants involved, while others express their intent to continue working on the problem based on the discussion. There is no explicit consensus on the best approach yet.
Contextual Notes
Participants are navigating the definitions and units associated with the formulas, and there is mention of the need for the temperature to be in Kelvin for one of the equations. The original poster has also indicated a reliance on reputable sources for the molar mass of methane.
Similar threads
- · Replies 5 ·
- · Replies 14 ·
- · Replies 12 ·
- · Replies 6 ·
- · Replies 3 ·
- · Replies 6 ·