Tclack
- 37
- 0
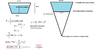
So I'm in the Navy. I work on an aircraft carrier that just went into drydock. The drydock has a large trapezoid door keeping the water out on one side. Our captain wants to know how much force is being held back on that door. I found the dimensions of it and calculated it, but now I'm having doubts.
My work is in the attached document.
I DID keep in mind that the water only goes up 50 feet (though 60ft is the trapezoid's height)
In the end, I got a force of 12.9 million lbs. When I divide by the area of the door (8162sq.ft) I get an average pressure of ~11psi...
14.7 psi is atmospheric pressure telling me that my answer is wrong, but I can't see why! Any thoughts?
My work is in the attached document.
I DID keep in mind that the water only goes up 50 feet (though 60ft is the trapezoid's height)
In the end, I got a force of 12.9 million lbs. When I divide by the area of the door (8162sq.ft) I get an average pressure of ~11psi...
14.7 psi is atmospheric pressure telling me that my answer is wrong, but I can't see why! Any thoughts?