ameeno97
- 34
- 0
Hi guys
I have a question regarding the working principle of gas burner. I was always have this question " How gas burner works using gas pressure lower -after been regulated- than atmospheric air ? "
Won't the the atmospheric air pressure prevent the gas from flowing?
Gas Burners
Both natural gas and propane burners work on the same principle called the "venturi effect". It says that as a gas or fluid passes through a pipe that narrows or widens, the velocity and pressure of the gas or fluid vary. As the pipe narrows, the gas flows more rapidly. What sounds like a surprise but holds true, is that when the fluid or gas flows faster through the narrow sections, the pressure actually decreases rather than increases. The venturi tube is a large diameter tube, gradually feeding into a smaller tube and then gradually becoming a larger tube
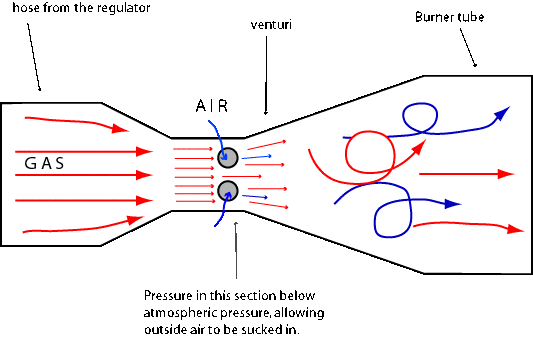
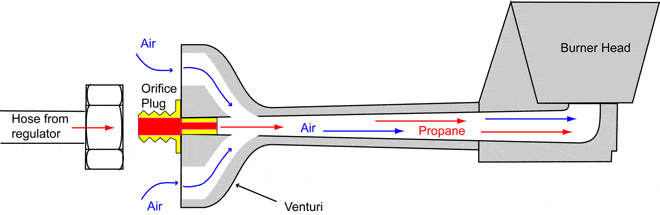
On the photos below there are examples of different burners, all operating on a venturi principle. One of the main advantages of Venturi systems is operation without electricity


The most important part of the burner is the orifice plug with the hole in it. This is the point where the gas escapes from the hose or a pipe and enters the mixing bell of the burner. Orifice plugs are replaceable and screwed into the orifice spud. Nearly all atmospheric (venturi) burners have a gas orifice that is accurately fixed in the burner throat providing air intake. The hole in the orifice is very small to provide the correct gas flow and to provide sufficient velocity to ensure there is a suction (vacuum) available for the correct air inspiration.
The question again is " When the gas start to flow from the regulator with say 2 PSI, Why the existing atmospheric air pressure (14.6 PSI) just prevent the gas from flowing?
I hope that you get my question right
I have a question regarding the working principle of gas burner. I was always have this question " How gas burner works using gas pressure lower -after been regulated- than atmospheric air ? "
Won't the the atmospheric air pressure prevent the gas from flowing?
Gas Burners
Both natural gas and propane burners work on the same principle called the "venturi effect". It says that as a gas or fluid passes through a pipe that narrows or widens, the velocity and pressure of the gas or fluid vary. As the pipe narrows, the gas flows more rapidly. What sounds like a surprise but holds true, is that when the fluid or gas flows faster through the narrow sections, the pressure actually decreases rather than increases. The venturi tube is a large diameter tube, gradually feeding into a smaller tube and then gradually becoming a larger tube
On the photos below there are examples of different burners, all operating on a venturi principle. One of the main advantages of Venturi systems is operation without electricity
The most important part of the burner is the orifice plug with the hole in it. This is the point where the gas escapes from the hose or a pipe and enters the mixing bell of the burner. Orifice plugs are replaceable and screwed into the orifice spud. Nearly all atmospheric (venturi) burners have a gas orifice that is accurately fixed in the burner throat providing air intake. The hole in the orifice is very small to provide the correct gas flow and to provide sufficient velocity to ensure there is a suction (vacuum) available for the correct air inspiration.
The question again is " When the gas start to flow from the regulator with say 2 PSI, Why the existing atmospheric air pressure (14.6 PSI) just prevent the gas from flowing?
I hope that you get my question right
Last edited: