Discussion Overview
The discussion centers on the Kinetic Theory of gases, specifically focusing on the concept of particle collisions and the associated probability functions. Participants explore the meaning of the velocity distribution function and its implications for understanding collisions between gas molecules.
Discussion Character
- Exploratory
- Technical explanation
- Conceptual clarification
- Debate/contested
Main Points Raised
- Some participants question whether ##f(v,v')## represents the velocity distribution function and if it indicates the fraction of molecules with specific velocities.
- Others clarify that ##f(v,v')## is the probability of two molecules with velocities ##\vec v## and ##\vec v'## colliding, noting potential confusion due to its expression over a small range of velocities.
- There is a query about the derivation of the exponential expression for ##f(v^2)## and ##f(v'^2)##, and how the product of these functions relates to the probability of collision.
- One participant suggests replacing "f" with "p" for clarity and provides a mathematical expression related to the distribution of kinetic energies.
- Concerns are raised about the interpretation of the product of ##f(v)## and ##f(v')## as implying a molecule can possess two velocities simultaneously, which seems nonsensical to some participants.
- Questions arise regarding the relationship between collision time, probability of collision, mean free path, and the distribution of velocities.
Areas of Agreement / Disagreement
Participants express differing views on the interpretation of the probability functions and their implications for collisions. There is no consensus on the relationship between the probability of collision and the distribution of velocities, leaving the discussion unresolved.
Contextual Notes
Some participants note that the discussion lacks clarity on the relationship between collision time, relative velocities, and the probability functions, indicating potential limitations in the current understanding.
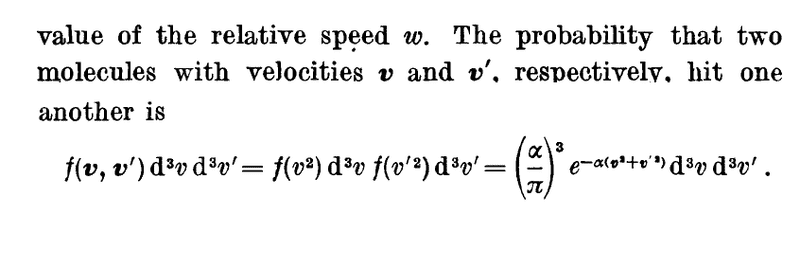 What is f(v,v') here? Is it the velocity distribution function? If so, isn't it simply the fraction of molecules with velocities v and v' ?
What is f(v,v') here? Is it the velocity distribution function? If so, isn't it simply the fraction of molecules with velocities v and v' ?