Discussion Overview
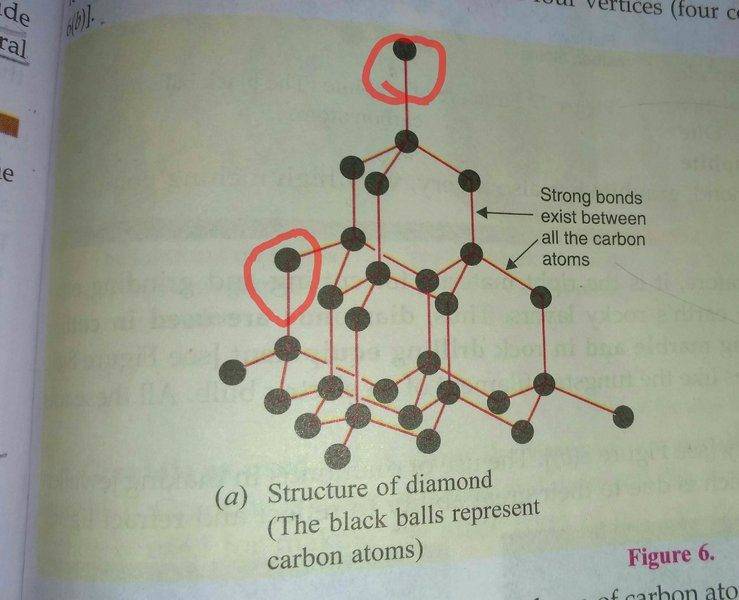
The discussion revolves around the accuracy of a diagram depicting the structure of diamond, specifically addressing the bonding of carbon atoms within the crystal lattice. Participants explore the implications of surface atoms versus interior atoms, conductivity, and the representation of atomic bonds in diagrams.
Discussion Character
- Debate/contested
- Technical explanation
- Conceptual clarification
Main Points Raised
- One participant questions the accuracy of a textbook diagram, noting that some carbon atoms appear to be bonded to fewer than four others.
- Another participant clarifies that the diagram represents only a portion of the diamond structure, suggesting that the missing atoms are not depicted but exist in the full crystal.
- It is noted that surface atoms have fewer neighbors than those inside the crystal, leading to questions about the implications for electrical conductivity.
- Some participants argue that surface atoms are bonded to other elements like hydrogen or oxygen, which may affect conductivity.
- There is a discussion about the nature of "free" electrons and their role in conductivity, emphasizing that not all surface electrons contribute to electrical conduction.
- Participants express uncertainty about the concept of the conduction band and its relevance to the discussion of diamond's conductivity.
- One participant mentions the possibility of dangling bonds on the diamond surface, indicating that not all surfaces are the same in terms of bonding characteristics.
Areas of Agreement / Disagreement
Participants generally agree that the diagram does not represent the entire structure of diamond and that surface atoms behave differently than interior atoms. However, there remains disagreement regarding the implications of surface bonding on conductivity and the nature of free electrons.
Contextual Notes
Some participants express confusion about the conduction band and the conditions necessary for conductivity, indicating a need for further clarification on these concepts. The discussion also touches on the limitations of visual representations in conveying complex atomic structures.
Who May Find This Useful
This discussion may be useful for high school students studying chemistry and physics, particularly those interested in atomic structure, bonding, and electrical properties of materials.