jrklx250s
- 15
- 0
Homework Statement
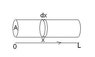
Given the temperature of an ideal gas in a tub where it varies linearly from (x=0) to (x=L)
T = To + [(TL - To)/L]x
where To is temperature at 0 length and TL is the temperature at the end of the tube
We are suppose to show that the equation of state for this ideal gas is
PV=nR[(TL-To)/ln(TL/To)]
As well as if then To = TL = T show that the equation of state reduces to the obvious one...PV=nRT
The Attempt at a Solution
So first off I wasnt sure of where to start because I know the only changing variable in this question is temperature so I know we are suppose to rearrange this equation
T = To + [(TL - To)/L]x
However I'm not sure on how to derive this equation.
Mostly this is alegbra/dervatives/integration problem.
And I'm honestly stuck as to where to go from the temperature equation
Any suggestions on where to begin would be greatly appreciative.