pkv
- 11
- 0
A though experiment:
We have two gas chambers at the same temperature and pressure, separated by a wall punctuated by very small pores. Let's say small enough to allow the pass of only a few gas molecules at a time (so, no complex interactions between gas molecules inside the pore).
These pores are designed to allow molecules passing from one chamber to the other easy, and hard or very hard in reverse.
This design (for the pore) comes to mind:
Non-imaging optics CEC
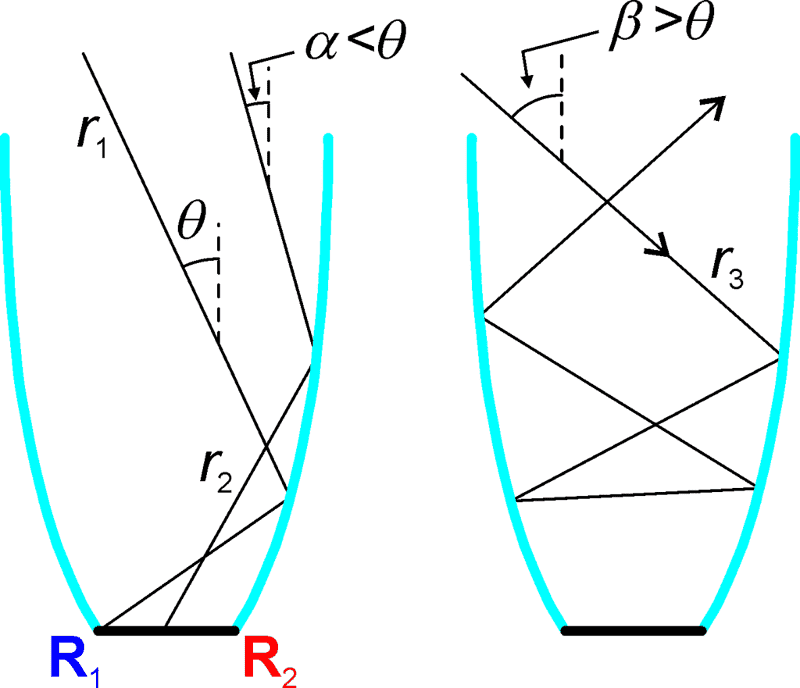
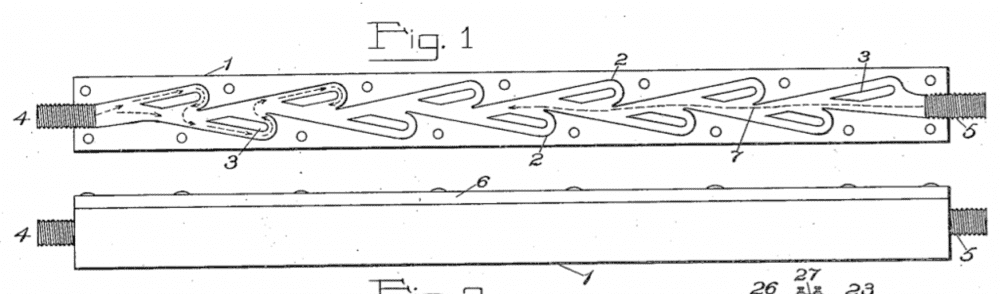
Another one could be a version of a Tesla valve, although I am not so sure of it's validity for single molecules.
 The question is: would pressure/temperature in both chambers remain stable, or would the gas slowly migrate to the "easy pass" chamber, raising it's pressure?
The question is: would pressure/temperature in both chambers remain stable, or would the gas slowly migrate to the "easy pass" chamber, raising it's pressure?
If the second option happens, the systems seems to me akin to the Maxwell demon, as it violates the second law.
Such a system would allow to extract mechanical energy from the gases without a cold reservoir, until it's kinetic energy is exhausted, so I guess it's impossible.
Where is the catch?
We have two gas chambers at the same temperature and pressure, separated by a wall punctuated by very small pores. Let's say small enough to allow the pass of only a few gas molecules at a time (so, no complex interactions between gas molecules inside the pore).
These pores are designed to allow molecules passing from one chamber to the other easy, and hard or very hard in reverse.
This design (for the pore) comes to mind:
Non-imaging optics CEC
Another one could be a version of a Tesla valve, although I am not so sure of it's validity for single molecules.
If the second option happens, the systems seems to me akin to the Maxwell demon, as it violates the second law.
Such a system would allow to extract mechanical energy from the gases without a cold reservoir, until it's kinetic energy is exhausted, so I guess it's impossible.
Where is the catch?