Discussion Overview
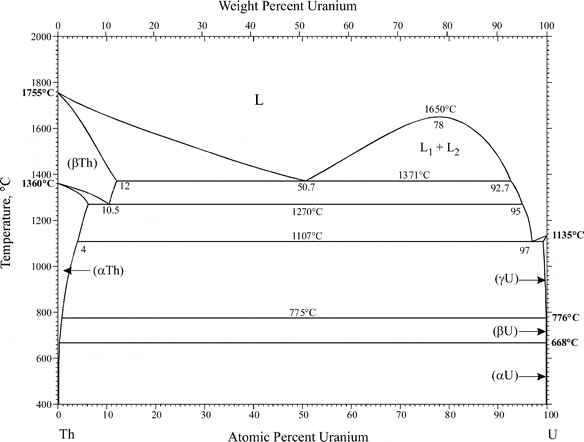
The discussion revolves around the behavior of thorium at very low concentrations in a two-component alloy, specifically in the context of uranium casting. Participants explore the mechanisms of fractionation and phase behavior when thorium is present at parts per billion or parts per million levels.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
Main Points Raised
- One participant questions the mechanisms driving thorium's fractionation during uranium casting, noting that material scientists suggest low concentrations are too dilute to form separate phases.
- Another participant suggests that the issue may relate more to partition coefficients rather than phase diagrams, indicating that thorium may have a higher affinity for "slag" than for uranium.
- A further reply proposes that fractionation could occur through random contact due to convection or induction mixing, suggesting that thorium has a high partition coefficient for materials other than uranium.
- One participant cautions against conflating "sticking to something else" with "phase-separating from uranium," arguing that interactions can occur even at low concentrations where phase separation is not expected.
Areas of Agreement / Disagreement
Participants express differing views on the relevance of phase separation versus partition coefficients in explaining thorium's behavior. There is no consensus on the mechanisms at play or the implications of low concentration on phase behavior.
Contextual Notes
The discussion highlights the complexity of interactions at low concentrations and the potential for different interpretations of phase behavior versus partitioning, without resolving the underlying assumptions or definitions involved.
Who May Find This Useful
Researchers and practitioners in materials science, particularly those focused on alloy behavior, phase diagrams, and the effects of trace elements in metallurgical processes.