Discussion Overview
The discussion revolves around the phenomenon of carryover cooking, specifically focusing on the mathematical modeling of how the internal temperature of meat continues to rise after being removed from a heat source. Participants explore various theoretical approaches, the physical and chemical processes involved, and the implications of thermal conductivity in this context.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
- Mathematical reasoning
Main Points Raised
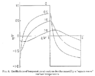
- One participant proposes a simple mathematical model using three regions (center, outer layer, and ambient) and Newton's Cooling Law to demonstrate carryover cooking.
- Another participant emphasizes the importance of thermal conductivity in understanding the temperature gradient and heat migration within the meat.
- Some participants suggest that carryover cooking involves both physical processes and chemical reactions, with discussions on the breakdown of organic structures and heat transfer through liquids.
- A later reply challenges the notion that carryover cooking is not a physical process, asserting that it is indeed driven by thermal conductivity and heat transfer principles.
- Several participants mention the existence of online simulators and models, such as one from MIT, that incorporate carryover cooking into their calculations, although the specifics of these models are not fully explored.
- One participant expresses a desire for a simpler conceptual explanation to convince skeptics of carryover cooking, arguing that the temperature gradient alone suffices to explain the phenomenon.
- Another participant suggests simplifying experimental setups to measure thermal conductivity and internal temperature changes more effectively.
Areas of Agreement / Disagreement
Participants express differing views on the complexity of modeling carryover cooking, with some advocating for simpler explanations while others delve into more technical discussions. There is no consensus on a single model or approach, and the discussion remains unresolved regarding the best way to represent the phenomenon.
Contextual Notes
Participants note the limitations of their models, including the dependence on assumptions about thermal conductivity and the complexity of the physical and chemical processes involved. Some suggest that existing models may not fully capture the nuances of carryover cooking.