Maximusflash
Hey
I've gotten some good answers here before, so I'm giving it another try:
Recently I attended a building-engineering meeting, and trying to learn something new (or refresh old knowledge in some cases..) I had a hydrodynamic question/case. My firm is engineering a concrete tank for a water treatment center, so the loads on the main construction are known. But I was curious about forces on a couple of valves:
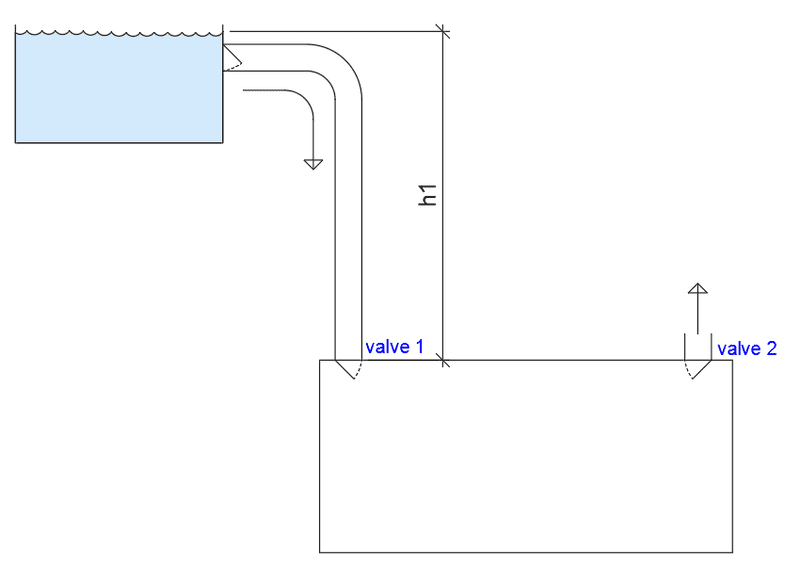
Now, the pressure in the bottom tank is given by the height from the water level into top tank, density of fluid and g. So in the case were bottom tank, top tank and tubes are full, valve 2 closed, the pressure at valve 1 would be h1 x ρ x g. This would be the case wether valve 1 is open or closed. Valve 1 would have the same pressure over it as under it, and opening/closing wouldn't change anything.
Now if everything has filled up, you close valve 1 (nothing happens). But as you open valve 2 things change. Pressure goes from h1 x ρ x g to nothing? Reference height is now equal to water height. Water will not explode out of the valve (depending on how solid the tank is: if it is elastic I guess some water could run out as it decompresses?). But at valve 1 it now has higher pressure at the top than the bottom. So we close valve 2 again, and valve 1 still has the h1 pressure at the top and nothing under. Water can not be compressed, so to even out the pressure one has to some how add pressure to the tank, or open valve 1. This will be "hard" as it has a high pressure on top of it. Could valve 1 be sort of a "spring" that compresses down to the bottom tank by the force/pressure on top of it, and equalizing the pressure before opening?
I've gotten some good answers here before, so I'm giving it another try:
Recently I attended a building-engineering meeting, and trying to learn something new (or refresh old knowledge in some cases..) I had a hydrodynamic question/case. My firm is engineering a concrete tank for a water treatment center, so the loads on the main construction are known. But I was curious about forces on a couple of valves:
Now, the pressure in the bottom tank is given by the height from the water level into top tank, density of fluid and g. So in the case were bottom tank, top tank and tubes are full, valve 2 closed, the pressure at valve 1 would be h1 x ρ x g. This would be the case wether valve 1 is open or closed. Valve 1 would have the same pressure over it as under it, and opening/closing wouldn't change anything.
Now if everything has filled up, you close valve 1 (nothing happens). But as you open valve 2 things change. Pressure goes from h1 x ρ x g to nothing? Reference height is now equal to water height. Water will not explode out of the valve (depending on how solid the tank is: if it is elastic I guess some water could run out as it decompresses?). But at valve 1 it now has higher pressure at the top than the bottom. So we close valve 2 again, and valve 1 still has the h1 pressure at the top and nothing under. Water can not be compressed, so to even out the pressure one has to some how add pressure to the tank, or open valve 1. This will be "hard" as it has a high pressure on top of it. Could valve 1 be sort of a "spring" that compresses down to the bottom tank by the force/pressure on top of it, and equalizing the pressure before opening?