Homework Help Overview
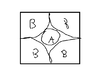
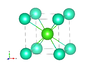
The discussion revolves around a problem related to cubic crystal structures, specifically focusing on the parameters and calculations involving the radius of ions in a CsCl structure. Participants are examining the relationships between geometric dimensions and ionic radii.
Discussion Character
- Exploratory, Assumption checking, Conceptual clarification
Approaches and Questions Raised
- Participants discuss methods for calculating the radius of ion A by utilizing the diagonal of a square and the geometry of the cubic structure. There are questions about the number of nearest neighbors and the implications of different crystal structures.
Discussion Status
Some participants have provided insights and hints regarding the calculations and assumptions about the crystal structure. There is an ongoing exploration of the correct parameters and relationships, with no explicit consensus reached yet.
Contextual Notes
There are indications of confusion regarding the correct values for the radius and the assumptions about the crystal structure, particularly in relation to the CsCl model. The original poster's calculations differ from expected results, prompting further discussion.