Discussion Overview
The discussion revolves around the workings of a simple voltaic cell as described by Alessandro Volta, including the materials used, the assumptions made about electrode charges, and the historical context of terminology. Participants explore the principles of electrochemistry, the nature of electrode reactions, and the confusion arising from different sources regarding the polarity of electrodes.
Discussion Character
- Exploratory
- Debate/contested
- Technical explanation
- Conceptual clarification
Main Points Raised
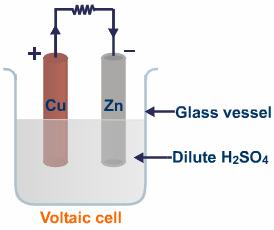
- Some participants describe Volta's setup involving a glass electrolytic cell filled with dilute sulfuric acid and electrodes made of copper and zinc, questioning how it functions without an external power source.
- Others explain that a chemical reaction occurs at the electrodes, generating an electromotive force, and assert that a voltaic cell operates independently as a battery.
- There is a discussion about the historical context of the terms "positive" and "negative" charges, with some arguing that Volta's terminology was arbitrary and could have been different.
- One participant challenges the accuracy of a textbook claim regarding the use of a light bulb or galvanometer in Volta's experiments, noting that these devices were not available during his time.
- Some participants express confusion over the differing claims about the polarity of the electrodes, with one stating that zinc is negatively charged due to its ionization potential, while another references sources that contradict this view.
- A participant mentions that their textbook states the electromotive force of the cell is 1.10 V, which aligns with their understanding but conflicts with other sources.
- There is a request for clarification on the conflicting information regarding electrode polarity, with some sources indicating that zinc is positive and copper negative, which is contrary to what the participant's textbook states.
Areas of Agreement / Disagreement
Participants express disagreement regarding the polarity of the electrodes and the historical accuracy of Volta's experimental setup. Multiple competing views remain on the interpretation of the electrochemical principles involved, and the discussion does not reach a consensus.
Contextual Notes
Participants note limitations in their references, including the potential for mixed information from various sources and the historical context of terminology used in early electrochemistry. The discussion highlights the confusion stemming from different interpretations of electrode charge and the evolution of scientific understanding.
Who May Find This Useful
This discussion may be useful for students studying electrochemistry, educators seeking to understand historical perspectives on electrical terminology, and anyone interested in the foundational concepts of voltaic cells and their operation.