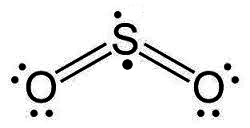
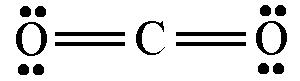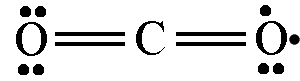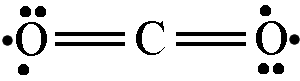Discussion Overview
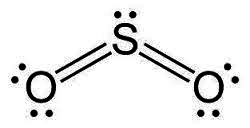
The discussion revolves around the representation of lone pairs in Lewis dot structures, questioning why unbonded electrons are typically drawn in pairs despite the potential for electron repulsion. Participants explore the implications of electron pairing, the validity of alternative drawings, and the conventions used in chemical notation.
Discussion Character
- Exploratory
- Debate/contested
- Conceptual clarification
Main Points Raised
- Some participants suggest that electrons tend to pair up due to spin coupling within their orbitals, which may explain the convention of drawing them as pairs.
- Others argue that the diagrams are merely notational and do not accurately represent the distribution of electrons in a molecule.
- A participant questions whether all proposed drawings are valid and if they can be used interchangeably.
- One participant explains that for elements in the first period, there are limitations on the number of available valence orbitals, which influences how electrons are paired.
- Another participant indicates that while alternative drawings can be made, they may not adhere to established conventions and could lead to misunderstandings.
- There is a suggestion that there is no single accurate notation, as different notations serve different purposes depending on what is being emphasized.
Areas of Agreement / Disagreement
Participants express differing views on the validity of alternative drawings and the reasons behind the convention of pairing electrons. The discussion remains unresolved regarding the best representation of lone pairs and the implications of electron pairing.
Contextual Notes
Limitations include the dependence on definitions of orbitals and the varying interpretations of electron pairing in different contexts. The discussion does not resolve the mathematical or conceptual complexities involved in electron configurations.
Who May Find This Useful
This discussion may be of interest to high school students learning about Lewis dot structures, educators seeking to understand student misconceptions, and individuals exploring the conventions of chemical notation.