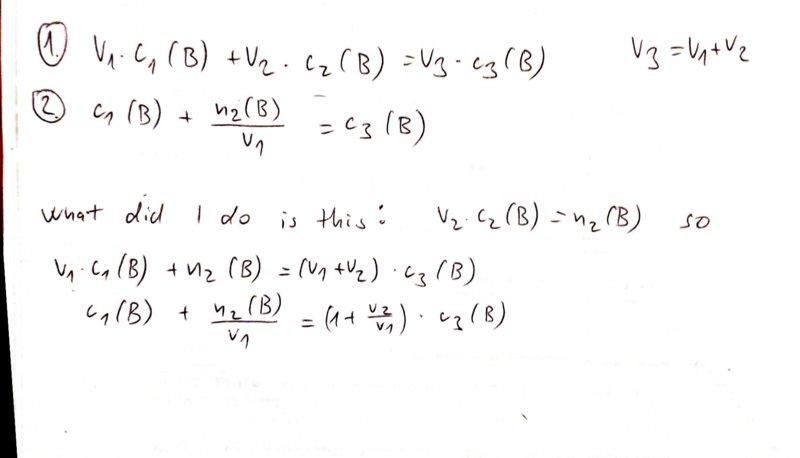Discussion Overview
The discussion revolves around the validity of an equation related to adding pure substances to a solution, specifically addressing the conditions under which this equation holds true. Participants explore the implications of volume changes when adding solutes to solutions, focusing on dilute versus concentrated solutions.
Discussion Character
- Technical explanation
- Conceptual clarification
- Debate/contested
Main Points Raised
- One participant questions the correctness of an equation and seeks clarification on the disappearance of a term in the equation.
- Another participant suggests that if V2 is much lower than V1, the term (1+V2/V1) can be approximated as 1, indicating a potential simplification in the equation.
- A participant clarifies that the equation in question pertains to adding pure B to a solution, where the assumption is that the volume of the solution does not change significantly when adding a small amount of solute.
- Further inquiry arises about why the volume does not change when adding pure B, prompting a discussion on the effects of dilution.
- It is noted that while the volume does change, it may be negligible in dilute solutions, and that for precise calculations, volume changes should be considered, especially in concentrated solutions.
Areas of Agreement / Disagreement
Participants express differing views on the significance of volume changes when adding solutes, with some agreeing that the change can be negligible in dilute solutions, while others emphasize the importance of accounting for volume changes in more concentrated scenarios. The discussion remains unresolved regarding the specific conditions under which the original equation is valid.
Contextual Notes
Limitations include the assumption that the volume change is negligible in dilute solutions, which may not hold true for concentrated solutions. The discussion also reflects varying interpretations of the equation's applicability based on the concentration of the solutions involved.