Homework Help Overview
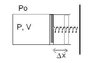
The discussion revolves around calculating the work done by an ideal gas on a Hooke's spring when heat is added, resulting in a tripling of both volume and pressure. The problem involves concepts from thermodynamics and mechanics, particularly the relationship between gas behavior and spring dynamics.
Discussion Character
Approaches and Questions Raised
- Participants explore the relationship between the work done on the spring and the work done by the gas, questioning how to relate mechanical work to thermodynamic work. There are attempts to derive equations involving pressure, volume, and the spring constant.
Discussion Status
Several participants are actively engaging with the problem, offering insights and raising questions about assumptions, such as the initial conditions of the spring and gas. Some have proposed equations but express uncertainty about how to proceed, indicating a lack of consensus on the approach to take.
Contextual Notes
Participants note the absence of specific values for the spring constant and the displacement of the spring, which complicates the calculations. There is also discussion about the nature of the thermodynamic process involved, with participants unsure whether it fits standard classifications like isothermal or adiabatic.