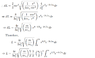Discussion Overview
The discussion revolves around calculating the moment of inertia of an electron in a hydrogen atom using classical and quantum mechanical approaches, particularly focusing on the implications of the wavefunction and Planck's constant. Participants explore the relationship between classical mechanics and quantum mechanics in this context.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
- Mathematical reasoning
Main Points Raised
- One participant discusses the calculation of spin for an electron using classical mechanics, questioning the appropriateness of using Bohr radius and the implications of electron cloud distribution.
- Another participant seeks assistance in calculating the moment of inertia of an electron using the wavefunction, indicating uncertainty in their results.
- There is a request for clarification on the purpose of the calculations and the theoretical formulas being used.
- A participant references a related question about approximating spin and the challenges of using a spherical model for the electron.
- One participant attempts to connect the calculation of energy in NMR to classical reasoning, raising questions about the derivation of spin values and their implications.
- Another participant calculates orbital angular momentum classically and compares it to the quantum mechanical value of h-bar.
- Concerns are raised about the validity of the wavefunction and the applicability of Planck's constant to massive particles like electrons, suggesting a potential misunderstanding of its role in quantum mechanics.
- A participant expresses the desire to avoid circular reasoning when discussing Planck's constant and its relation to classical mechanics and quantum mechanics.
- One participant asserts that proving the existence of Planck's constant through classical reasoning is not feasible, suggesting that coincidences do not equate to proof.
Areas of Agreement / Disagreement
Participants express differing views on the validity of using classical mechanics to derive quantum properties, particularly regarding the wavefunction and Planck's constant. There is no consensus on the appropriateness of the methods discussed or the interpretations of the results.
Contextual Notes
Participants highlight limitations in their approaches, including the reliance on classical models for quantum phenomena, the challenges of defining appropriate parameters like radius for calculations, and the potential for circular reasoning in their derivations.