SUMMARY
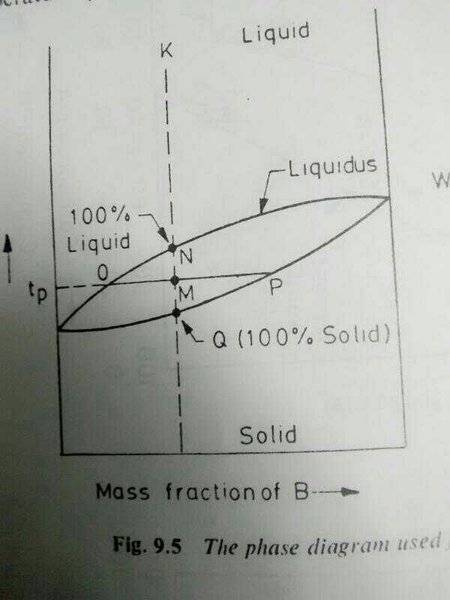
The discussion clarifies the application of the Lever Rule in determining the amounts of liquid and solid phases in a cooling mixture. Specifically, it addresses the confusion regarding the identification of the liquidus and solidus lines in phase diagrams, as illustrated in Fig 3 from Tulane University's website. The example provided demonstrates how to calculate the fractions of liquid (L) and solid (S) using the equations LxBliq + SxBsol = xB and L + S = 1, confirming the validity of the Lever Rule.
PREREQUISITES
- Understanding of phase diagrams and their components
- Familiarity with the concepts of liquidus and solidus lines
- Basic algebra for solving equations
- Knowledge of concentration terms in materials science
NEXT STEPS
- Study phase diagrams in materials science
- Learn about the Lever Rule and its applications in thermodynamics
- Explore the concept of liquidus and solidus lines in detail
- Practice solving problems involving the Lever Rule and phase compositions
USEFUL FOR
This discussion is beneficial for materials scientists, chemical engineers, and students studying phase equilibria and thermodynamics, particularly those interested in the behavior of mixtures during cooling processes.