JoeyF
- 13
- 0
- TL;DR
- I would like to understand how much water condenses from a fixed volume of steam as a result of cooling the system to ambient. Starting Saturated Steam mixture has a pressure of 18 psi in a volume of 2.46e-5 m^3
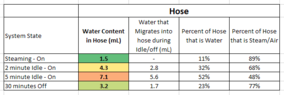
I've developed a Steam Cleaner using a through-flow heater (800W) . Solenoid pump is roughly 60 mL/min . Heater Power is limited via Thermostat. The Pump is always running. The Steam Outlet is controlled by a deadman-style valve (normally closed). The Steam outlet is also on a hose that has roughly 6 feet of tubing (I estimate about 2.46 e-5 m^3 of volume containing steam during use). It is a closed system (with a limiting pressure relief valve that wastes water pre-heater when the outlet valve is closed. Works great! However...
Since it is a closed system, after I power down the machine, the leftover steam condenses inside the system. I would like to be able to calculate the amount of Condensate (in grams/mL) that will condense and get leftover in my system. The downside to this is a big slug of water that comes out of the cleaner upon subsequent use. SOME of this liquid is a consequence of the large temperature swings I see in my heater due to the relatively large tolerance on my Thermostat and lack of flowrate controls, but I'm consistently measuring around 16 mL of leftover water during subsequent uses. I want to understand how much liquid water is actually from the Steam condensing. Can anybody help me understand a principle/equation that might get me started?
Some more supporting information: Operating Pressure ~18 psi. After System is turned off, System drops to ~ (-)8 psi.
Thanks!
Since it is a closed system, after I power down the machine, the leftover steam condenses inside the system. I would like to be able to calculate the amount of Condensate (in grams/mL) that will condense and get leftover in my system. The downside to this is a big slug of water that comes out of the cleaner upon subsequent use. SOME of this liquid is a consequence of the large temperature swings I see in my heater due to the relatively large tolerance on my Thermostat and lack of flowrate controls, but I'm consistently measuring around 16 mL of leftover water during subsequent uses. I want to understand how much liquid water is actually from the Steam condensing. Can anybody help me understand a principle/equation that might get me started?
Some more supporting information: Operating Pressure ~18 psi. After System is turned off, System drops to ~ (-)8 psi.
Thanks!