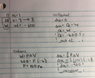SUMMARY
The discussion centers on the determination of internal energy changes in a diatomic gas, specifically when given a specific heat capacity (Cp) of 7/2. The user questions whether this indicates a diatomic gas, which would alter the equation for internal energy. The correct approach involves using the equation dU=(3/2)nRdT for monoatomic gases, while for diatomic gases, the equation becomes dU=(5/2)nRdT. The conclusion drawn is that the distinction between monoatomic and diatomic is not necessary for solving the problem, as the first law of thermodynamics provides a clear path to the solution.
PREREQUISITES
- Understanding of the first law of thermodynamics
- Familiarity with specific heat capacities (Cp and Cv)
- Knowledge of ideal gas behavior
- Ability to manipulate thermodynamic equations
NEXT STEPS
- Study the derivation of the first law of thermodynamics
- Learn about the differences between monoatomic and diatomic gases
- Explore the implications of specific heat capacities on internal energy
- Investigate the relationship between enthalpy (ΔH) and internal energy (ΔU)
USEFUL FOR
Students studying thermodynamics, physics educators, and anyone looking to deepen their understanding of internal energy changes in gases.