Manganese
- 9
- 0
Hello,
I'm a chemist and I'm currently working on my PhD on materials science.
I'm trying to determine the pressure difference necessary to achieve the desired gas flow inside a chamber. The scheme of the chamber is the following:
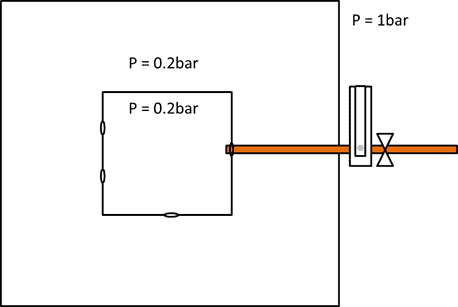
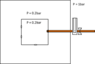
I have a big chamber which is evacuated to 0.2 bar, and another small one inside with 3 holes (Diameter = 6 mm). There is a copper pipe connecting the small chamber to the outside environment (air, P = 1bar) and regulated by a valve. Mass flow is read with a flowmeter. When the valve is open, air is forced inside the chamber but the internal pressure of the chamber is kept constant (the pump is powerful enough to maintain 0.2 bar). The small chamber is not air tight and there are many gaps between the wall, so I don't think evacuating it should be an issue (and so far it wasn't).
For a ΔP = 0.8 bar, the flowmeter reads a volumetric gas flow of 5 L/min, but I want to calculate the maximum gas flow I can achieve at a specified pressure difference between the inside and outside of the chamber. I know I could just toy around with the pressure inside and make a table, but I want to calculate it from theory. The specifics are the following:
μ = 1.8⋅10-5 Kg/ms
D = 0.003 m
Pout = 1 bar
T = 298 K
ρ = 1.225⋅10-3Kg/L
For Pin = 0.2 bar, the flowmeter reads Ṽ = 5 L/min, thus:
ṁ = Ṽ*ρ = 5⋅1.225⋅10-3/60 = 1.03⋅10-4 Kg/s
ṁ = ρvπD2/4 ⇒ v = 4ṁ/(ρvπD2)
Re = ρvD/μ ⇒ Re = 4ṁ/(μπD) = 2429
I don't know if these data are necessary or useful at all for what I want to find out, since in my case ṁ is technically unknown, but this is what I was able to calculate. How can I get the dependence of ṁ from ΔP? Any equation I missed along the way?
Thanks
I'm a chemist and I'm currently working on my PhD on materials science.
I'm trying to determine the pressure difference necessary to achieve the desired gas flow inside a chamber. The scheme of the chamber is the following:
I have a big chamber which is evacuated to 0.2 bar, and another small one inside with 3 holes (Diameter = 6 mm). There is a copper pipe connecting the small chamber to the outside environment (air, P = 1bar) and regulated by a valve. Mass flow is read with a flowmeter. When the valve is open, air is forced inside the chamber but the internal pressure of the chamber is kept constant (the pump is powerful enough to maintain 0.2 bar). The small chamber is not air tight and there are many gaps between the wall, so I don't think evacuating it should be an issue (and so far it wasn't).
For a ΔP = 0.8 bar, the flowmeter reads a volumetric gas flow of 5 L/min, but I want to calculate the maximum gas flow I can achieve at a specified pressure difference between the inside and outside of the chamber. I know I could just toy around with the pressure inside and make a table, but I want to calculate it from theory. The specifics are the following:
μ = 1.8⋅10-5 Kg/ms
D = 0.003 m
Pout = 1 bar
T = 298 K
ρ = 1.225⋅10-3Kg/L
For Pin = 0.2 bar, the flowmeter reads Ṽ = 5 L/min, thus:
ṁ = Ṽ*ρ = 5⋅1.225⋅10-3/60 = 1.03⋅10-4 Kg/s
ṁ = ρvπD2/4 ⇒ v = 4ṁ/(ρvπD2)
Re = ρvD/μ ⇒ Re = 4ṁ/(μπD) = 2429
I don't know if these data are necessary or useful at all for what I want to find out, since in my case ṁ is technically unknown, but this is what I was able to calculate. How can I get the dependence of ṁ from ΔP? Any equation I missed along the way?
Thanks