Discussion Overview
The discussion revolves around the efficiency of the reversible Otto cycle compared to the Carnot cycle, focusing on the conditions under which these efficiencies are defined and the implications of operating between different temperature reservoirs. Participants explore theoretical aspects of thermodynamic cycles, particularly in the context of reversible engines.
Discussion Character
- Debate/contested
- Technical explanation
- Conceptual clarification
Main Points Raised
- Some participants assert that every reversible engine operating between the same temperatures should have the same efficiency as the Carnot engine, leading to confusion regarding the efficiency of the Otto cycle.
- Others clarify that the Carnot efficiency applies specifically to engines operating between two heat reservoirs, and that the Otto cycle operates under different conditions, resulting in a different efficiency formula.
- There is a contention regarding the statement that reversible engines do not necessarily have the same efficiencies, with some participants referencing the Carnot cycle as an upper limit for efficiency in classical thermodynamic engines.
- One participant reflects on the implications of reversing cycles and the necessity of operating between two heat reservoirs for the Carnot efficiency to apply, noting that this cannot be generalized to all scenarios.
- Another participant questions the mechanics of reversing a cycle, suggesting that it involves work done on the engine to transfer heat from a lower to a higher temperature reservoir.
- A later reply discusses a proof involving two reversible engines and the transfer of heat, concluding that the efficiency argument holds only for two reservoirs and cannot be generalized.
Areas of Agreement / Disagreement
Participants express differing views on the relationship between the efficiencies of reversible engines and the conditions under which the Carnot efficiency applies. There is no consensus on the implications of these efficiencies when considering different temperature reservoirs.
Contextual Notes
Participants highlight limitations in the applicability of the Carnot efficiency to the Otto cycle, emphasizing the need for specific conditions (i.e., operating between two heat reservoirs) for the Carnot theorem to hold. The discussion reveals a complexity in understanding the efficiency of different thermodynamic cycles.
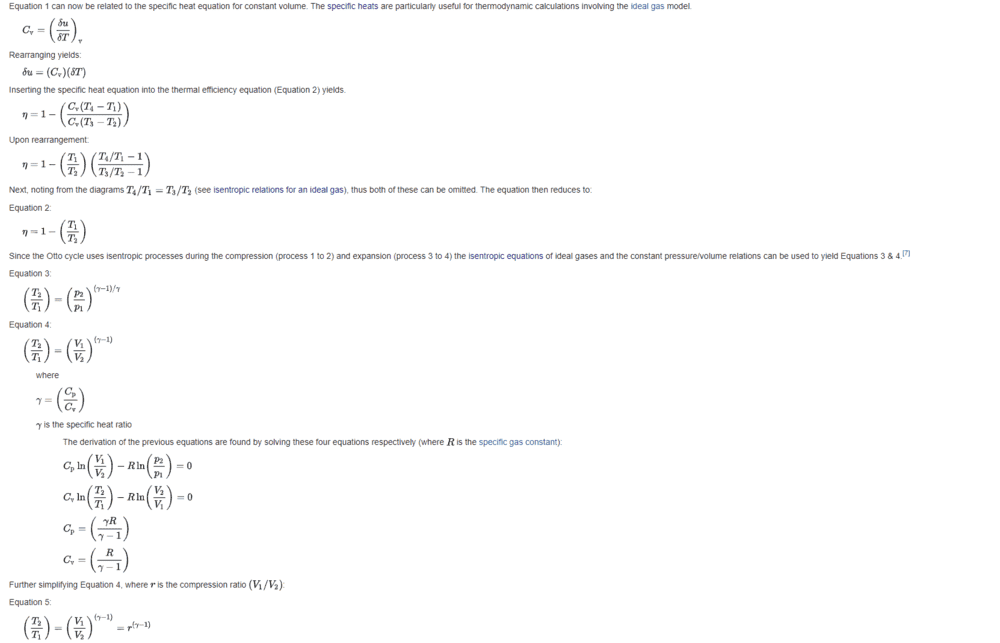
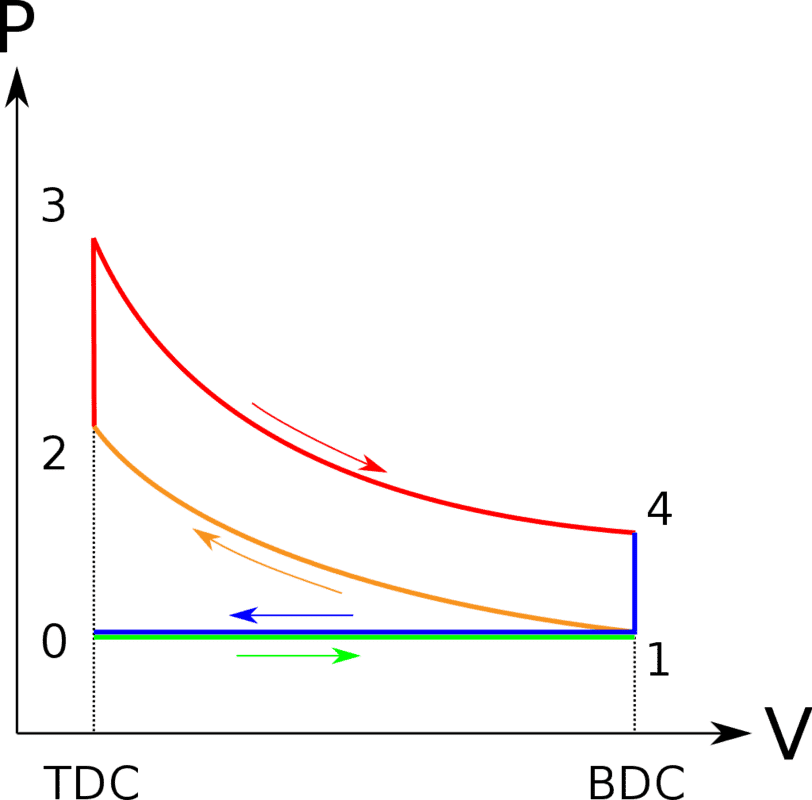 https://en.wikipedia.org/wiki/Otto_cycle
https://en.wikipedia.org/wiki/Otto_cycle