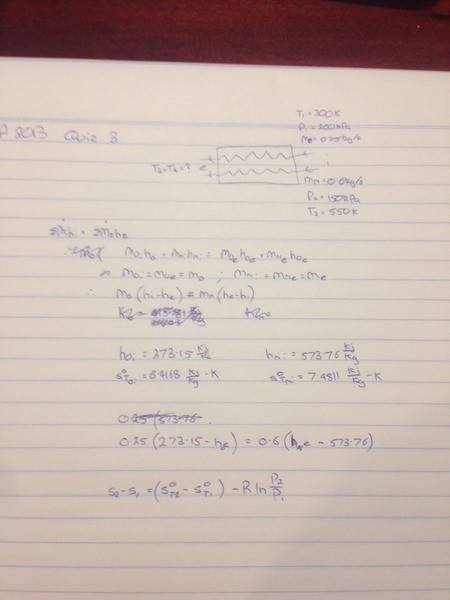SUMMARY
The discussion centers on calculating entropy change in a co-current heat exchanger involving oxygen and nitrogen streams. Participants emphasize the need to equate the enthalpy increase of the oxygen stream with the enthalpy decrease of the nitrogen stream to determine the exit temperatures. The entropy change per kilogram for both gases can be calculated using the formula ΔS = Cp * ln(T2/T1), where Cp is the specific heat capacity, and T1 and T2 are the initial and final temperatures, respectively. The negligible pressure drop allows for a simplified analysis focused solely on temperature changes.
PREREQUISITES
- Understanding of thermodynamic principles, specifically heat exchangers.
- Familiarity with the concept of entropy and its calculation.
- Knowledge of specific heat capacities for gases, particularly oxygen and nitrogen.
- Basic skills in logarithmic functions and their application in thermodynamic equations.
NEXT STEPS
- Study the calculation of entropy change using the formula ΔS = Cp * ln(T2/T1).
- Research the specific heat capacities of oxygen and nitrogen at varying temperatures.
- Explore the principles of co-current and counter-current heat exchangers.
- Learn about the impact of pressure on thermodynamic properties in gas mixtures.
USEFUL FOR
Students and professionals in thermodynamics, mechanical engineers, and anyone involved in heat exchanger design and analysis will benefit from this discussion.