jaumzaum
- 433
- 33
I was studying wind formation from the continent to the sea during day (direct circulation) and the explanation I found in almost all the textbooks was that:
1. In the continent during day the ground becomes hotter
2. The particles in the air increases their temperatures due to induction
3. When the air increases its temperature, the particles become far apart from each other, and the density reduces
4. Due to buoyancy, this air rises
5. As the air rises, for some reason, this creates a low pressure site at the ground and a high pressure site at the sky
6. The air now flows from the sky of the continent (high pressure) to the sky of the ocean (low pressure)
7. To complete the cycle the air drops (as it cools down) to the surface of the ocean creating a high pressure point at sea surface
8. Then it comes back to the continent
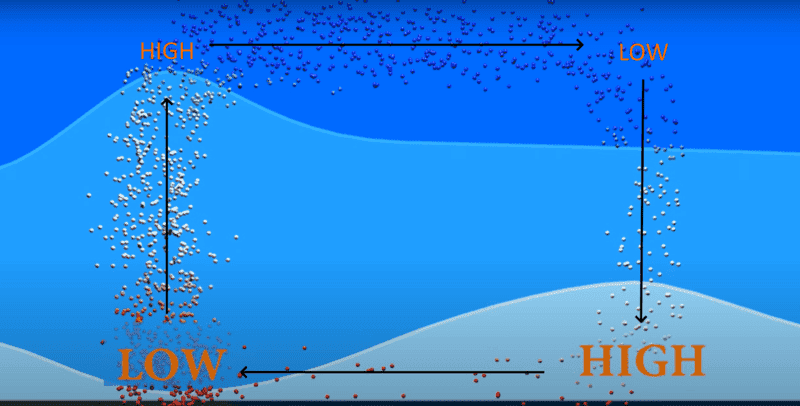
I can understand all the 8 statements, except the fifth. Why is the continent ground low pressure?
I mean, if we use PV=nRT or better PM = dRT (where M is molar mass and d is density), I know that when T increases d will decrease. But consider T doubles. If d drops less than 50% than P will increase. If d drops 50% P will remain constant. If d drops more than 50% P will decrease. So why the density at the ground drops always more than the increase in temperature, this is not obvious to me. Less density air will have lower pressure, but hotter air will have higher pressure, so the question is, why the first event wins?
Thank you!
1. In the continent during day the ground becomes hotter
2. The particles in the air increases their temperatures due to induction
3. When the air increases its temperature, the particles become far apart from each other, and the density reduces
4. Due to buoyancy, this air rises
5. As the air rises, for some reason, this creates a low pressure site at the ground and a high pressure site at the sky
6. The air now flows from the sky of the continent (high pressure) to the sky of the ocean (low pressure)
7. To complete the cycle the air drops (as it cools down) to the surface of the ocean creating a high pressure point at sea surface
8. Then it comes back to the continent
I can understand all the 8 statements, except the fifth. Why is the continent ground low pressure?
I mean, if we use PV=nRT or better PM = dRT (where M is molar mass and d is density), I know that when T increases d will decrease. But consider T doubles. If d drops less than 50% than P will increase. If d drops 50% P will remain constant. If d drops more than 50% P will decrease. So why the density at the ground drops always more than the increase in temperature, this is not obvious to me. Less density air will have lower pressure, but hotter air will have higher pressure, so the question is, why the first event wins?
Thank you!