Discussion Overview
The discussion centers around a new antiviral strategy developed by researchers at MIT Lincoln Laboratory, specifically focusing on a class of agents called DRACOs (Double-stranded RNA Activated Caspase Oligomerizers) that aim to target a broad range of viral infections. The conversation explores the theoretical underpinnings, potential applications, and limitations of this approach, including its effectiveness against various viruses and the mechanisms involved in viral detection and response.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
Main Points Raised
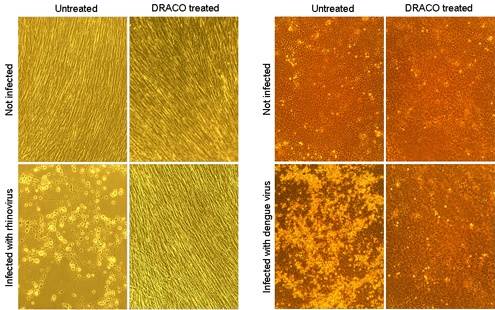
- Some participants highlight that DRACOs target long double-stranded RNA (dsRNA) produced during viral replication, which is typically absent in healthy mammalian cells, suggesting a potential mechanism for viral detection.
- Concerns are raised about the effectiveness of DRACOs in vivo, particularly regarding their stability and delivery to infected tissues, as well as the potential for viruses to evade detection mechanisms.
- One participant argues that DRACOs do not exclusively rely on long dsRNA for pathogen recognition, suggesting that various detection and effector domains could be utilized, including those that may target retroviruses like HIV.
- Another participant mentions that mammalian cells already possess mechanisms to detect dsRNA, such as through TLR-3 receptors, which induce interferon production as part of the immune response.
- There is a reiteration of the concern that while DRACOs show promise, their application against all virus types, particularly retroviruses, may not be feasible due to the specific nature of dsRNA detection.
Areas of Agreement / Disagreement
Participants express differing views on the mechanisms and effectiveness of DRACOs, particularly regarding their ability to target retroviruses and the reliance on dsRNA for detection. The discussion remains unresolved, with multiple competing perspectives on the potential and limitations of the DRACO approach.
Contextual Notes
Participants note limitations in the current understanding of DRACOs, including the need for further research to validate their effectiveness in clinical settings and the complexities of viral evasion strategies that may impact the proposed mechanisms.