Westlund2
- 7
- 0
Are these cis/trans isomers? Why or why not?
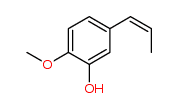
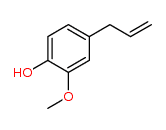
The discussion revolves around the identification of cis/trans isomers in certain molecules. Participants explore the criteria that define cis/trans isomerism, including structural considerations and the implications of ring structures.
Participants do not appear to reach a consensus on whether the molecules are cis/trans isomers, as differing viewpoints on the criteria and structural implications are presented.
The discussion lacks detailed definitions of cis/trans isomerism and does not clarify the specific structural features of the molecules being analyzed, which may affect the conclusions drawn.
espen180 said:Let's hear your take on it first.