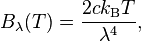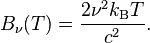Discussion Overview
The discussion revolves around the measurement of spectral radiance of blackbodies, specifically addressing the differences in peak intensities when measured in terms of wavelength versus frequency. Participants explore the implications of these differences and the equipment used for such measurements.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
Main Points Raised
- Some participants question whether two types of equipment are necessary to measure spectral radiance for Bλ and Bν, suggesting that one measurement method could suffice due to the relationship between frequency and wavelength.
- Others argue that different peak values for wavelength and frequency can be obtained by maximizing the corresponding distribution functions, although the physical significance of these peaks is debated.
- Some participants assert that the formulas provided by the original poster may be incorrect or not representative of the actual blackbody spectrum, leading to confusion about the existence of peaks.
- There is a discussion about the shape of spectral density intensity functions in wavelength versus frequency space, with some noting that while the peaks occur at different locations, the energy between two wavelengths remains consistent across both representations.
- One participant mentions the use of a tungsten filament light bulb as a calibration source for measuring spectral radiance, detailing the experimental setup involving a diffraction grating and photodiode.
- Several participants emphasize the need for correct blackbody formulas, with some suggesting that the original formulas may represent approximations rather than complete functions.
- There is a contention regarding the physical measurability of peak values, with some asserting that they can be measured while others argue that the peaks may not hold significant physical meaning.
Areas of Agreement / Disagreement
Participants express differing views on the necessity of distinct measurement instruments for wavelength and frequency, the correctness of the formulas presented, and the physical significance of the peak values. The discussion remains unresolved with multiple competing perspectives.
Contextual Notes
Some formulas referenced may not accurately represent the blackbody spectrum, and there are indications that participants are working with approximations or earlier versions of these formulas. The discussion also highlights the complexity of converting between wavelength and frequency representations.