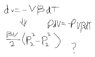Discussion Overview
The discussion revolves around estimating the pressure increase required to impart 1 J of mechanical work in the reversible compression of 1 mol of silver and alumina at room temperature. Participants explore the application of relevant equations and seek clarification on unit conversions and assumptions related to the problem.
Discussion Character
- Homework-related
- Mathematical reasoning
Main Points Raised
- One participant presents the problem statement and equations related to mechanical work and volume changes, expressing difficulty in finding a solution.
- Another participant confirms the correctness of the equation involving pressure and suggests substituting numerical values, assuming a starting pressure of 1 atm.
- A participant inquires whether the same equation can be applied to part (b) of the problem, noting that it does not specify mechanical work or reversibility.
- There is a request for a solution to the problem, which is met with a reminder of forum rules against providing direct solutions.
- A participant raises a question about necessary unit conversions for molar volume and mentions discrepancies in beta values for silver, seeking clarification on the differences in molar volumes provided.
Areas of Agreement / Disagreement
Participants generally agree on the use of the equation for calculating pressure increase but express uncertainty regarding its application to different parts of the problem and the need for unit conversions. The discussion remains unresolved regarding the specific numerical answers and the application of the equations.
Contextual Notes
Participants mention various assumptions, such as the starting pressure and the nature of the work being done, but these assumptions are not universally agreed upon. There is also uncertainty regarding the appropriate unit conversions and the interpretation of different molar volumes.