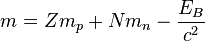SUMMARY
The discussion centers on the Liquid Drop Model and its implications for nuclear mass calculations. It clarifies that the mass of the nucleus is derived from the equation where the binding energy is subtracted, leading to the conclusion that the mass of the nucleus is less than the sum of the individual nucleon masses. This is due to the energy required to separate nucleons, which is accounted for by the binding energy. The participants emphasize the importance of understanding energy conservation in nuclear physics.
PREREQUISITES
- Understanding of the Liquid Drop Model in nuclear physics
- Familiarity with binding energy concepts
- Basic knowledge of nucleon composition (protons and neutrons)
- Concept of mass-energy equivalence (E=mc²)
NEXT STEPS
- Research the Liquid Drop Model and its applications in nuclear physics
- Explore the concept of binding energy and its calculation
- Study mass-energy equivalence and its implications in nuclear reactions
- Learn about the role of nucleons in atomic structure and stability
USEFUL FOR
Students and professionals in nuclear physics, physicists studying atomic structure, and anyone interested in the principles of mass-energy relationships in nuclear reactions.