Discussion Overview
The discussion revolves around the nature of sunlight, specifically its absorption spectrum and the implications for the perception of color. Participants explore the relationship between wavelengths and colors, the significance of absorption lines in the solar spectrum, and the definition of "white light." The conversation includes both theoretical and conceptual aspects of light and color perception.
Discussion Character
- Exploratory
- Conceptual clarification
- Debate/contested
- Technical explanation
Main Points Raised
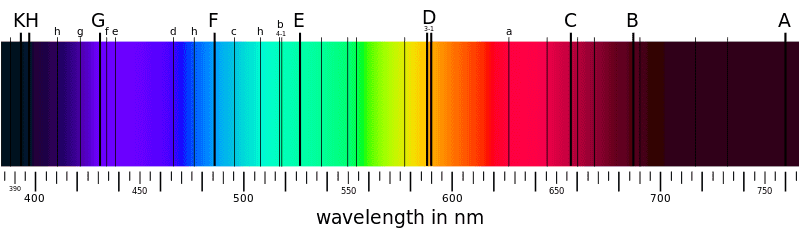
- Some participants propose that the black lines in the sunlight absorption spectrum indicate that certain wavelengths are missing, which could imply that sunlight does not contain all colors.
- Others argue that the presence of absorption lines does not mean that those wavelengths are entirely absent; rather, there is just a reduction in intensity at those wavelengths.
- A participant questions the distinction between 'all wavelengths' and 'all colors,' seeking clarification on what it means to perceive light as white.
- There is a discussion about whether specific wavelengths, such as 650 nm, can be considered a color and how combinations of wavelengths contribute to color perception.
- Some participants note that even at absorption wavelengths, there is still some light present, albeit at a lower intensity compared to adjacent wavelengths.
- Technical details are provided regarding specific absorption lines, including those associated with sodium and hydrogen, and their relevance to the discussion of the solar spectrum.
Areas of Agreement / Disagreement
Participants express differing views on the implications of absorption lines in the solar spectrum. There is no consensus on whether the presence of these lines means sunlight lacks certain colors or wavelengths, and the discussion remains unresolved regarding the definitions of color and white light.
Contextual Notes
Participants acknowledge the complexity of the relationship between wavelengths and colors, and the discussion highlights the dependence on definitions and interpretations of light perception. There are also unresolved questions about how absorption lines are represented in recordings and their implications for understanding sunlight.