SUMMARY
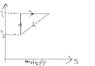
The discussion centers on calculating heat input and efficiency for a temperature-entropy (T-S) cyclic process. The efficiency η is expressed as η=(Q1/Q2)-1=(T1/T2)-1. The heat extracted during the linear process II-III is calculated as Q_{II-III} = -\frac{1}{2\alpha} (T_1^2 - T_2^2), indicating heat extraction. Additionally, the process III-I is identified as adiabatic with Q_{III-I}=0, while the isothermal process I-II requires further analysis of entropy change.
PREREQUISITES
- Understanding of thermodynamic cycles
- Familiarity with temperature-entropy (T-S) diagrams
- Knowledge of heat transfer equations
- Basic calculus for integration and differentiation
NEXT STEPS
- Study the derivation of heat transfer in isothermal processes
- Explore the implications of adiabatic processes in thermodynamics
- Learn about the Carnot cycle and its efficiency calculations
- Investigate the relationship between entropy and heat in cyclic processes
USEFUL FOR
Students and professionals in thermodynamics, mechanical engineers, and anyone involved in energy systems analysis will benefit from this discussion.