Discussion Overview
The discussion revolves around the relationship between temperature and pressure for water, particularly in the context of a solar collector project. Participants explore thermodynamic processes and seek to establish accurate values for pressure and temperature in a controlled volume system involving water and molten salt.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
- Mathematical reasoning
Main Points Raised
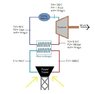
- One participant seeks an equation relating temperature and pressure for water at constant volume, aiming to confirm the accuracy of their values for a solar collector project.
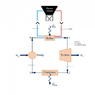
- Questions arise regarding the fluids involved in the system, particularly the pressure values of 100 bar before the turbine and 1.5 bar after, which some participants find unrealistic.
- Another participant notes that at 510°C, water behaves similarly to an ideal gas, and discusses the implications of phase changes between liquid and gas water, suggesting the use of a phase diagram for better understanding.
- There is a repeated emphasis on the need for realistic values for pressure and temperature based on the phase diagram, with acknowledgment that liquid water is essentially incompressible.
- Participants express confusion about determining values for different segments of the system, particularly regarding mass flow rate, temperature, pressure, work of the pump, turbine, and heat transfer in the heat exchanger.
- One participant mentions that mass flow rate remains constant if there is no accumulation, but notes insufficient information to determine pressure and temperature accurately.
- References to the Carnot engine are made, suggesting it as a theoretical framework for understanding efficiency limits in the system.
Areas of Agreement / Disagreement
Participants do not reach a consensus on the accuracy of the proposed pressure values or the methods to determine the necessary thermodynamic parameters. Multiple competing views and uncertainties remain regarding the relationship between temperature and pressure in the context of the project.
Contextual Notes
Participants highlight limitations in the available information, particularly regarding the assumptions needed to accurately determine pressure and temperature values. The discussion also indicates a dependence on phase diagrams and the complexities of the system setup.