Discussion Overview
The discussion revolves around the behavior of phases along the triple line for water, focusing on the definitions of 'quality' and the existence of solid, liquid, and vapor phases at specific points along the line. Participants explore theoretical and conceptual aspects of phase transitions and equilibrium conditions.
Discussion Character
- Exploratory
- Conceptual clarification
- Debate/contested
Main Points Raised
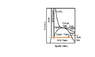
- Some participants question whether 'quality' is defined along the triple line and what fractions of solid and liquid exist at various points.
- At point C, some assert there is pure vapor, while at point A, there is pure solid, and at point B, there may be pure liquid or a mixture of solid and vapor depending on pressure.
- One participant emphasizes that being "exactly" at point B is not possible, as any slight increase in pressure results in pure liquid, while a slight decrease leads to a mixture of solid and vapor.
- Another participant raises the issue of determining the proportions of solid and liquid between points B and C, suggesting that latent heat plays a role in phase changes without necessarily having all three phases coexisting.
- There is a suggestion that the concept of 'quality' may not be valid along the triple line due to the non-uniqueness of phase combinations at various points.
- Some participants agree that between points A and B, solid must be present, while between B and C, gas must be present, but the exact phases at point B remain debated.
- Quantification of phases using tie lines and the lever rule is mentioned as a method to determine proportions of solid, liquid, and vapor at various points.
Areas of Agreement / Disagreement
Participants express multiple competing views regarding the existence and proportions of phases at specific points along the triple line, particularly at point B. The discussion remains unresolved regarding the applicability of 'quality' and the exact nature of phase presence at various pressures.
Contextual Notes
There are limitations in defining 'quality' along the triple line, and the discussion highlights dependencies on pressure and temperature conditions, as well as the role of latent heat in phase transitions.