- #1
MathewsMD
- 433
- 7
When using L'Hospital's rule to differentiate this function, why does the second term have a negative 1 multiplying it? Is it not already negative? Is it because of the -h in the expression?
Since the limit is on h, that's the variable used in differentiation. So, yes, the -1 comes about from differentiating f(x - h) with respect to h.MathewsMD said: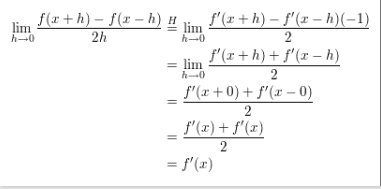
When using L'Hospital's rule to differentiate this function, why does the second term have a negative 1 multiplying it? Is it not already negative? Is it because of the -h in the expression?
Negative 1 is used in mathematical equations because it represents the additive inverse or opposite of 1. This means that when negative 1 is added to 1, the result is 0. It is an important concept in mathematics and is used in various calculations and proofs.
In physics, negative 1 is used to represent the direction of motion or force in the opposite direction. It is also used in the concept of negative acceleration, where an object's velocity decreases over time. Negative 1 is an important factor in many equations and formulas in physics.
In electricity, negative 1 is used to indicate a negative charge. It is also used in the concept of electrical potential, where a negative value represents a decrease in potential energy. Negative 1 is an important factor in understanding the behavior and interactions of electric charges.
In computer programming, negative 1 is used to represent the boolean value "false." This means that a statement or condition is not true or does not exist. It is also used in binary code to represent the negative or "off" state for a particular bit. Negative 1 is an essential component in creating logic and decision-making processes in programming.
In chemistry, negative 1 is used to indicate the charge of an ion. For example, if an atom gains one electron, it will have a negative 1 charge. Negative 1 is also used in oxidation-reduction reactions, where it represents the loss of one electron by an atom. This concept is crucial in understanding chemical reactions and the behavior of atoms and molecules.