Homework Help Overview
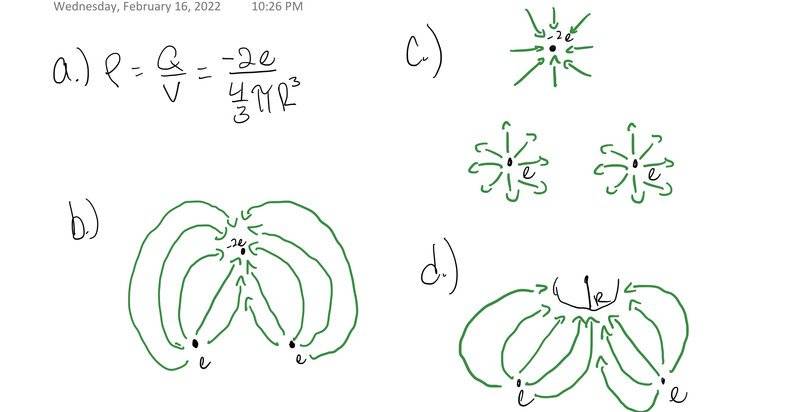
The discussion revolves around the electric field lines of a water molecule (H2O), focusing on the modeling of the oxygen atom as a conducting sphere and the implications of charge distribution in relation to electric field lines and charge density functions.
Discussion Character
- Exploratory, Assumption checking, Conceptual clarification
Approaches and Questions Raised
- Participants discuss the integration of volume charge density and its implications, questioning the uniformity of charge distribution over the conducting sphere. There are inquiries about the behavior of electric field lines in relation to the charges present, particularly concerning the distribution of charge and the use of Dirac delta functions.
Discussion Status
Participants are actively engaging with the problem, offering corrections and suggestions for clarification. There is a focus on understanding the distribution of charge and the relationship between electric field lines and enclosed charge, with some guidance provided on drawing Gaussian surfaces.
Contextual Notes
There are mentions of specific parts of the problem (a, b, c, d) and the need for clarity regarding the treatment of protons and electrons, as well as the implications of charge density functions across different regions. The discussion reflects uncertainty about the correct application of concepts related to charge distribution and electric fields.