maajdl
Gold Member
- 391
- 29
Hello,
From wikipedia, this is the partition function for a "classical continuous system":
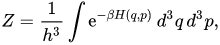
This is the pillar of classical statistical physics, but it can be seen as a mere kind of "mathematical transform" .
It can be used even without thinking to statistics or temperature.
If we focus only on the potential energy part of this integral, then
H = V(q)
is a function of q and the "positions of all other particles" of the system.
I question myself:
Would the full knowledge of Z(β) contain the full information about the "other particles".
And therefore, could the knowledge of Z(β) be traced back (inverted) to the positions of the atoms?
I hope this question doesn't look too fancyful.
I find it interresting because it would cast geometrical data in a 1-variable function Z(β) !
Thanks for your suggestions!
Michel
From wikipedia, this is the partition function for a "classical continuous system":
This is the pillar of classical statistical physics, but it can be seen as a mere kind of "mathematical transform" .
It can be used even without thinking to statistics or temperature.
If we focus only on the potential energy part of this integral, then
H = V(q)
is a function of q and the "positions of all other particles" of the system.
I question myself:
Would the full knowledge of Z(β) contain the full information about the "other particles".
And therefore, could the knowledge of Z(β) be traced back (inverted) to the positions of the atoms?
I hope this question doesn't look too fancyful.
I find it interresting because it would cast geometrical data in a 1-variable function Z(β) !
Thanks for your suggestions!
Michel