msteves
- 7
- 0
I have a long steel uninsulated cylinder filled with hydraulic fluid (let's say it's mineral oil), and I need to figure out how many barrel heaters to clamp onto it in the winter months to prevent the steel surface temperature from dropping under 40 degrees Fahrenheit. My question is, how do I set up this problem? I currently have three 1200-W heaters, and I know that under an ambient temperature of 25F, I get sensors reporting under 40F, so I know these were undersized. I've attached a couple figures I made to try to best represent the problem. The numbers I have been getting have not instilled confidence in my work. Insulating these is tricky, but I know I will need to at least insulate portions of them.
Side note: the oil is locked up, with very occasional spurts of new oil entering the system at 100F. I am just assuming the oil is isolated in the cylinder.
Thanks for any help you can provide.
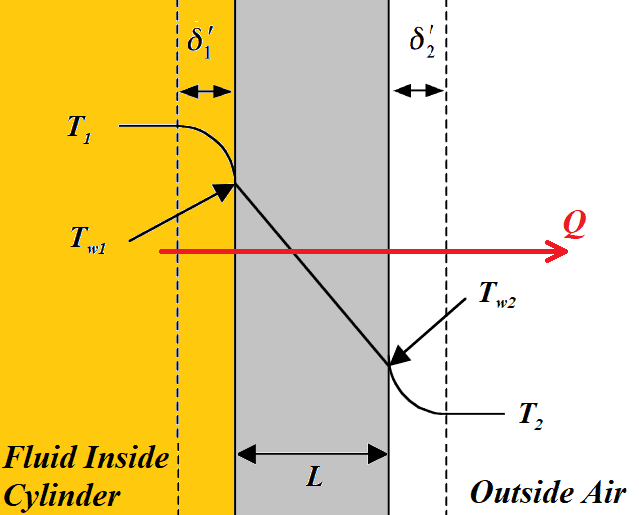
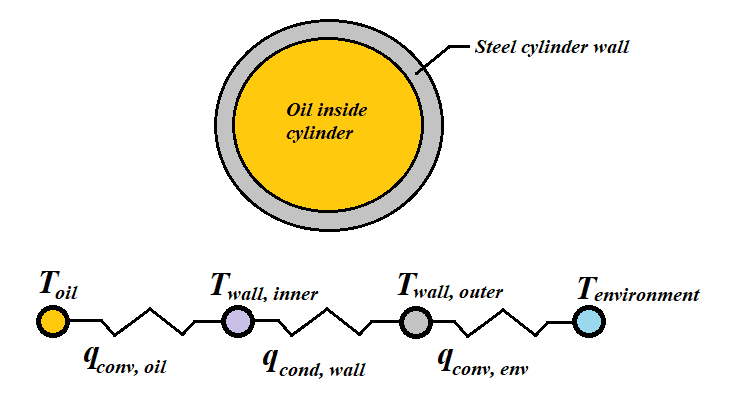
Side note: the oil is locked up, with very occasional spurts of new oil entering the system at 100F. I am just assuming the oil is isolated in the cylinder.
Thanks for any help you can provide.