Discussion Overview
The discussion centers around the search for a liquid with specific properties suitable for very low temperatures (around 100-150 K), including high density, high temperature of vaporization, low volatility, and high specific heat capacity. Participants explore potential applications and theoretical considerations related to thermal absorption and cryogenic cooling.
Discussion Character
- Exploratory
- Technical explanation
- Conceptual clarification
- Debate/contested
- Experimental/applied
Main Points Raised
- One participant seeks a liquid with high density, high vaporization temperature, low volatility, and high specific heat capacity for low-temperature applications.
- Another participant questions the vague terms used and suggests that clarifying the intended application could lead to better suggestions.
- A list of potential liquids with corresponding properties is provided, including hydrogen, fluorine, and various hydrocarbons, but no specific recommendations are made regarding their suitability for the stated requirements.
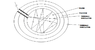
- A participant discusses the concept of a black body and proposes a system design involving a fluid within layers of thermal conductors and insulators to maximize thermal absorption.
- Concerns are raised about the practicality of using high boiling liquids near their freezing point and the implications of evaporation on thermal management.
- Another participant emphasizes the need for the fluid to have a high specific heat capacity to resist temperature increases and minimize evaporation.
- Questions are posed regarding the necessity and purpose of the proposed thermal absorption system.
- Discussion includes considerations about the design of the liquid-filled container and the effects of surface polishing on energy absorption and reflection.
- One participant suggests that allowing the liquid to evaporate could stabilize temperature and simplify measurements, questioning the need for a high thermal capacity fluid.
Areas of Agreement / Disagreement
Participants express various viewpoints on the properties and applications of the desired fluid, with no consensus reached on a specific solution or approach. The necessity and purpose of the thermal absorption system also remain contested.
Contextual Notes
Participants highlight limitations in the definitions of terms like "high" and "very," and the discussion reflects uncertainty regarding the feasibility of the proposed fluid properties and system design.